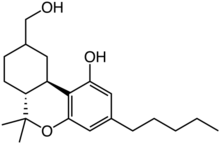
11-Hydroxyhexahydrocannabinol
11-Hydroxyhexahydrocannabinol (11-OH-9α-HHC and 11-OH-9β-HHC, or alternatively 7-OH-HHC under the monoterpenoid numbering system) is an active metabolite of tetrahydrocannabinol (THC) and the major metabolite of the trace cannabinoid hexahydrocannabinol (HHC).[1][2]
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C21H32O3 |
| Molar mass | 332.484 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
In a pathway that parallels the metabolism of the THC family of cannabinoids, following ingestion HHC undergoes hepatic metabolism by cytochrome p450 (predominantly the CYP3A4 isozyme, with some contribution from CYP2C9 and CYP2C19) to a multitude of oxygenated derivatives, including 8-OH-HHC and 11-OH-HHC. C11-oxidation is the major pathway of THC and HHC metabolism.[3]
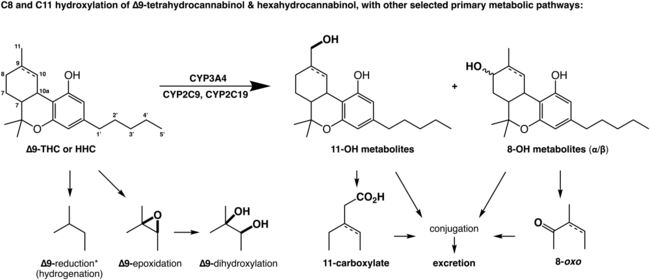
Like other 11-OH cannabinoid metabolites, 11-OH-9β-HHC retains activity comparable to HHC itself while the 9α-isomer is significantly less active.[4] However, upon formation it is rapidly metabolized further to the inactive 11-carboxylates, producing a shortened half-life within the body and lowering its bioavailability considerably through first-pass metabolism.[5][6]
The 11-OH-9β-HHC isomer is the structurally related methylene homologue of 11-Nor-9β-hydroxyhexahydrocannabinol also known as 9-Nor-9β-hydroxyhexahydrocannabinol.[7][8]
HU-243 is a synthetic analog of 11-OH-9β-HHC in which the natural n-pentyl side chain is replaced with a geminal-dimethylheptyl substitution. This significantly increases HU-243s binding affinity for the CB1 and CB2 receptors.[9]
See also
References
- Järbe TU, Hiltunen AJ, Lander N, Mechoulam R (August 1986). "Cannabimimetic activity (delta 1-THC cue) of cannabidiol monomethyl ether and two stereoisomeric hexahydrocannabinols in rats and pigeons". Pharmacology, Biochemistry, and Behavior. 25 (2): 393–399. doi:10.1016/0091-3057(86)90015-8. PMID 3020594. S2CID 28373651.
- Harvey DJ, Martin BR, Paton WD (1978). "Comparative in vivo Metabolism of Δ1-tetrahydrocannabinol (Δ1-THC), cannabidiol (CBD) and cannabinol (CBN) by Several Species". In Frigerio A (ed.). Recent Developments in Mass Spectrometry in Biochemistry and Medicine. Boston, MA: Springer US. pp. 161–184. doi:10.1007/978-1-4613-3991-5_13. ISBN 978-1-4613-3993-9.
- Dinis-Oliveira RJ (2016-01-02). "Metabolomics of Δ9-tetrahydrocannabinol: implications in toxicity". Drug Metabolism Reviews. 48 (1): 80–87. doi:10.3109/03602532.2015.1137307. PMID 26828228. S2CID 32913023.
- Mechoulam R, Lander N, Varkony TH, Kimmel I, Becker O, Ben-Zvi Z, et al. (October 1980). "Stereochemical requirements for cannabinoid activity". Journal of Medicinal Chemistry. 23 (10): 1068–1072. doi:10.1021/jm00184a002. PMID 7420350.
- Huestis MA (August 2007). "Human cannabinoid pharmacokinetics". Chemistry & Biodiversity. 4 (8): 1770–1804. doi:10.1002/cbdv.200790152. PMC 2689518. PMID 17712819.
- Lewis J, Molnar A, Allsop D, Copeland J, Fu S (January 2016). "Rapid elimination of Carboxy-THC in a cohort of chronic cannabis users". International Journal of Legal Medicine. 130 (1): 147–152. doi:10.1007/s00414-015-1241-z. PMID 26233612. S2CID 25318521.
- "9-Hydroxy-9-norhexahydrocannabinol". PubChem. U.S. National Library of Medicine.
- "7-Hydroxyhexahydrocannabinol". PubChem. U.S. National Library of Medicine.
- Devane WA, Breuer A, Sheskin T, Järbe TU, Eisen MS, Mechoulam R (May 1992). "A novel probe for the cannabinoid receptor". Journal of Medicinal Chemistry. 35 (11): 2065–2069. doi:10.1021/jm00089a018. PMID 1317925.