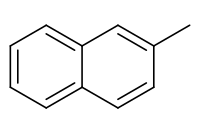
2-Methylnaphthalene
2-Methylnaphthalene is a polycyclic aromatic hydrocarbon (PAH).
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Methylnaphthalene | |
| Other names
β-methylnaphthalene | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.001.890 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C11H10 | |
| Molar mass | 142.201 g·mol−1 |
| -102.6·10−6 cm3/mol | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
On February 22, 2014, NASA announced a greatly upgraded database[1][2] for detecting and monitoring PAHs, including 2-methylnaphthalene, in the universe. According to NASA scientists, over 20% of the carbon in the universe may be associated with PAHs, possible starting materials for the formation of life.[1] PAHs seem to have been formed shortly after the Big Bang, are abundant in the universe,[3][4][5] and are associated with new stars and exoplanets.[1]
Several enzymes biodegrade 2-methyhlnaphthalene in anaerobic conditions.[6][7]
See also
References
- Hoover, Rachel (February 21, 2014). "Need to Track Organic Nano-Particles Across the Universe? NASA's Got an App for That". NASA. Retrieved February 22, 2014.
- Staff (October 29, 2013). "PAH IR Spectral Database". NASA. Retrieved March 12, 2014.
- Carey, Bjorn (October 18, 2005). "Life's Building Blocks 'Abundant in Space'". Space.com. Retrieved March 3, 2014.
- Hudgins, Douglas M.; Bauschlicher Jr, Charles W.; Allamandola, L. J. (October 10, 2005). "Variations in the Peak Position of the 6.2 μm Interstellar Emission Feature: A Tracer of N in the Interstellar Polycyclic Aromatic Hydrocarbon Population". Astrophysical Journal. 632: 316–332. doi:10.1086/432495.
- Allamandola, Louis; et al. (April 13, 2011). "Cosmic Distribution of Chemical Complexity". NASA. Archived from the original on February 27, 2014. Retrieved March 3, 2014.
- Meckenstock, Rainer U.; Manfred Böhm (2004), "Anaerobic degradation of polycyclic aromatic hydrocarbons", FEMS Microbiology Ecology, 49 (12): 27–36, doi:10.1016/j.femsec.2004.02.019
- Annweiler, Eva; Arne Materna (2000), "Anaerobic Degradation of 2-Methylnaphthalene by a Sulfate-Reducing Enrichment Culture", FEMS Microbiology Ecology, 66 (12): 5329–5333, doi:10.1128/AEM.66.12.5329-5333.2000, PMC 92464, PMID 11097910
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.