Oligodendrocyte
Oligodendrocytes (from Greek 'cells with a few branches'), also known as oligodendroglia, are a type of neuroglia whose main functions are to provide support and insulation to axons within the central nervous system (CNS) of jawed vertebrates. Their function is similar to that of Schwann cells, which perform the same task in the peripheral nervous system (PNS). Oligodendrocytes accomplish this by forming the myelin sheath around axons.[1] Unlike Schwann cells, a single oligodendrocyte can extend its processes to cover around 50 axons,[2] with each axon being wrapped in approximately 1 μm of myelin sheath. Furthermore, an oligodendrocyte can provide myelin segments for multiple adjacent axons.[1]
| Oligodendrocyte | |
|---|---|
 Oligodendrocytes form the electrical insulation around the axons of CNS nerve cells. | |
| Details | |
| Location | Central nervous system |
| Identifiers | |
| Latin | oligodendrocytus |
| MeSH | D009836 |
| TH | H2.00.06.2.00003, H2.00.06.2.01018 |
| FMA | 83665 54540, 83665 |
| Anatomical terms of microanatomy | |
Oligodendrocytes are exclusively found in the CNS, which comprises the brain and spinal cord. Initially, it was originally thought that these cells were produced in the ventral neural tube, the embryonic precursor to the CNS. However, recent research suggests that oligodendrocytes originate from the ventral ventricular zone of the embryonic spinal cord, with some potential concentrations in the forebrain.[3] Notably, oligodendrocytes are the last type of cell to be generated in the CNS.[4] Oligodendrocytes were discovered by Pío del Río Hortega.[5][6]
Classification
Oligodendrocytes are a type of glial cell. They arise during development from oligodendrocyte precursor cells (OPCs),[7] which can be identified by their expression of a number of antigens, including the ganglioside GD3,[8] the NG2 chondroitin sulfate proteoglycan, and the platelet-derived growth factor-alpha receptor subunit (PDGF-alphaR).[9] Mature oligodendrocytes are broadly classified into either myelinating or non-myelinating satellite oligodendrocytes. Precursors and both mature types are typically identified by their expression of the transcription factor OLIG2.[10]
Development
Most oligodendrocytes develop during embryogenesis and early postnatal life from restricted periventricular germinal regions.[11] Oligodendrocyte formation in the adult brain is associated with glial-restricted progenitor cells, known as oligodendrocyte progenitor cells (OPCs).[12] Subventricular zone cells migrate away from germinal[12] zones to populate both developing white and gray matter, where they differentiate and mature into myelin-forming oligodendrocytes.[13] However, it is not clear whether all oligodendrocyte progenitors undergo this sequence of events.
Between midgestation and term birth in human cerebral white matter, three successive stages of the classic human oligodendrocyte lineage are found: OPCs, immature oligodendrocytes (non-myelinating), and mature oligodendrocytes (myelinating).[14] It has been suggested that some undergo apoptosis[15] and others fail to differentiate into mature oligodendrocytes but persist as adult OPCs.[16] Remarkably, oligodendrocyte population originated in the subventricular zone can be dramatically expanded by administering epidermal growth factor (EGF).[17][18]
Function
Myelination
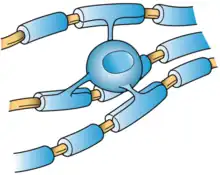
Mammalian nervous systems depend crucially on myelin sheaths, which reduce ion leakage and decrease the capacitance of the cell membrane, for rapid signal conduction.[19] Myelin also increases impulse speed, as saltatory propagation of action potentials occurs at the nodes of Ranvier in between Schwann cells (of the PNS) and oligodendrocytes (of the CNS). Furthermore, impulse speed of myelinated axons increases linearly with the axon diameter, whereas the impulse speed of unmyelinated cells increases only with the square root of the diameter. The insulation must be proportional to the diameter of the fibre inside. The optimal ratio of axon diameter divided by the total fiber diameter (which includes the myelin) is 0.6.[20]
Myelination is only prevalent in a few brain regions at birth and continues into adulthood. The entire process is not complete until about 25–30 years of age.[20] Myelination is an important component of intelligence, and white matter quantity may be positively correlated with IQ test results in children.[20] Rats that were raised in an enriched environment, which is known to increase cognitive flexibility, had more myelination in their corpus callosi.[21]
Metabolic support
Oligodendrocytes interact closely with nerve cells and provide trophic support by the production of glial cell line-derived neurotrophic factor (GDNF), brain-derived neurotrophic factor (BDNF), or insulin-like growth factor-1 (IGF-1).[22] They may also directly provide metabolites to neurons, as described by the lactate shuttle hypothesis.[23]
It is hypothesized that satellite oligodendrocytes (or perineuronal oligodendrocytes) are functionally distinct from other oligodendrocytes. They are not attached to neurons via myelin sheaths and, therefore, do not contribute to insulation. They remain opposed to neurons and regulate the extracellular fluid.[24] Satellite oligodendrocytes are considered to be a part of the grey matter whereas myelinating oligodendrocytes are a part of the white matter. They may support neuronal metabolism. Satellite oligodendrocytes may be recruited to produce new myelin after a demyelinating injury.[25]
Clinical significance
Diseases that result in injury to oligodendrocytes include demyelinating diseases such as multiple sclerosis and various leukodystrophies. Trauma to the body, e.g. spinal cord injury, can also cause demyelination. The immature oligodendrocytes, which increase in number during mid-gestation, are more vulnerable to hypoxic injury and are involved in periventricular leukomalacia.[26] This largely congenital condition of damage to the newly forming brain can therefore lead to cerebral palsy. In cerebral palsy, spinal cord injury, stroke and possibly multiple sclerosis, oligodendrocytes are thought to be damaged by excessive release of the neurotransmitter, glutamate.[27] Damage has also been shown to be mediated by N-methyl-D-aspartate receptors.[27] Oligodendrocyte dysfunction may also be implicated in the pathophysiology of schizophrenia and bipolar disorder.[28]
Oligodendrocytes are also susceptible to infection by the JC virus, which causes progressive multifocal leukoencephalopathy (PML), a condition that specifically affects white matter, typically in immunocompromised patients. Tumors of oligodendrocytes are called oligodendrogliomas. The chemotherapy agent Fluorouracil (5-FU) causes damage to the oligodendrocytes in mice, leading to both acute central nervous system (CNS) damage and progressively worsening delayed degeneration of the CNS.[29] [30] DNA methylation may also have a role in the degeneration of oligodendrocytes.[31]
See also
References
- Carlson, Neil (2010). Physiology of Behavior. Boston, MA: Allyn & Bacon. pp. 38–39. ISBN 978-0-205-66627-0.
- Baumann, Nicole; Pham-Dinh, Danielle (2001-04-01). "Biology of Oligodendrocyte and Myelin in the Mammalian Central Nervous System". Physiological Reviews. 81 (2): 871–927. doi:10.1152/physrev.2001.81.2.871. ISSN 0031-9333. PMID 11274346.
- Richardson, WD; Kessaris, N; Pringle, N (Jan 2006). "Oligodendrocyte wars". Nature Reviews. Neuroscience. 7 (1): 11–8. doi:10.1038/nrn1826. PMC 6328010. PMID 16371946.
- Thomas, JL; Spassky, N; Perez Villegas, EM; Olivier, C; Cobos, I; Goujet-Zalc, C; Martínez, S; Zalc, B (Feb 15, 2000). "Spatiotemporal development of oligodendrocytes in the embryonic brain". Journal of Neuroscience Research. 59 (4): 471–6. doi:10.1002/(SICI)1097-4547(20000215)59:4<471::AID-JNR1>3.0.CO;2-3. PMID 10679785. S2CID 42473842.
- Pérez-Cerdá, Fernando; Sánchez-Gómez, María Victoria; Matute, Carlos (2015). "Pío del Río Hortega and the discovery of the oligodendrocytes". Frontiers in Neuroanatomy. 9: 92. doi:10.3389/fnana.2015.00092. ISSN 1662-5129. PMC 4493393. PMID 26217196.
- James, Owen G; Mehta, Arpan R; Behari, Madhuri; Chandran, Siddharthan (June 2021). "Centenary of the oligodendrocyte". The Lancet Neurology. 20 (6): 422. doi:10.1016/S1474-4422(21)00136-8. PMC 7610932. PMID 34022167.
- Cameron-Curry, Patrizia; Le Douarin, Nicole M. (December 1995). "Oligodendrocyte precursors originate from both the dorsal and the ventral parts of the spinal cord". Neuron. 15 (6): 1299–1310. doi:10.1016/0896-6273(95)90009-8. PMID 8845154.
- Curtis et al., 1988; LeVine and Goldman, 1988; Hardy and Reynolds, 1991
- Pringle, NP; Mudhar, HS; Collarini, EJ; Richardson, WD (Jun 1992). "PDGF receptors in the rat CNS: during late neurogenesis, PDGF alpha-receptor expression appears to be restricted to glial cells of the oligodendrocyte lineage" (PDF). Development. 115 (2): 535–51. doi:10.1242/dev.115.2.535. PMID 1425339.
- Yokoo H, Nobusawa S, Takebayashi H, Ikenaka K, Isoda K, Kamiya M, Sasaki A, Hirato J, Nakazato Y (2004). "Anti-human Olig2 antibody as a useful immunohistochemical marker of normal oligodendrocytes and gliomas". Am J Pathol. 164 (5): 1717–25. doi:10.1016/S0002-9440(10)63730-3. PMC 1615653. PMID 15111318.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Vallstedt et al., 2004
- Menn, B; Garcia-Verdugo, JM; Yaschine, C; Gonzalez-Perez, O; Rowitch, D; Alvarez-Buylla, A (Jul 26, 2006). "Origin of oligodendrocytes in the subventricular zone of the adult brain". The Journal of Neuroscience. 26 (30): 7907–18. doi:10.1523/JNEUROSCI.1299-06.2006. PMC 6674207. PMID 16870736.
- Hardy and Reynolds, 1991; Levison and Goldman, 1993
- Barateiro, Andreia; Fernandes, Adelaide (September 2014). "Temporal oligodendrocyte lineage progression: In vitro models of proliferation, differentiation and myelination". Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. 1843 (9): 1917–1929. doi:10.1016/j.bbamcr.2014.04.018. PMID 24768715.
- Barres et al., 1992
- Wren et al., 1992
- Gonzalez-Perez, O, B; Romero-Rodriguez, R; Soriano-Navarro, M; Garcia-Verdugo, JM; Alvarez-Buylla, A (2009). "Epidermal growth factor induces the progeny of subventricular zone type B cells to migrate and differentiate into oligodendrocytes". Stem Cells. 27 (8): 2032–43. doi:10.1002/stem.119. PMC 3346259. PMID 19544429.
- Gonzalez-Perez, O, B; Alvarez-Buylla, A (Jun 24, 2011). "Oligodendrogenesis in the subventricular zone and the role of epidermal growth factor". Brain Research Reviews. 67 (1–2): 147–56. doi:10.1016/j.brainresrev.2011.01.001. PMC 3109119. PMID 21236296.
- Sokol, Stacey. "The Physiology and Pathophysiology of Multiple Sclerosis". Multiple Sclerosis: Physiological Tutorial. Archived from the original on 2012-07-16. Retrieved 2012-04-29.
- Fields, Douglas (18 February 2008). "White Matter Matters". Scientific American. 298 (March 2008): 54–61. Bibcode:2008SciAm.298c..54D. doi:10.1038/scientificamerican0308-54. PMID 18357821.
- Juraska J. M.; Kopcik J. R. (1988). "Sex and environmental influences on the size and ultrastructure of the rat corpus callosum". Brain Research. 450 (1–2): 1–8. doi:10.1016/0006-8993(88)91538-7. PMID 3401704. S2CID 2720782.
- Bradl (2010). "Oligodendrocytes: biology and pathology". Acta Neuropathol. 119 (1): 37–53. doi:10.1007/s00401-009-0601-5. PMC 2799635. PMID 19847447.
...oligodendrocytes can provide trophic support for neurons by the production of glial cell line-derived neurotrophic factor (GDNF), brain-derived neurotrophic factor (BDNF), or insulin-like growth factor-1 (IGF-1).
- Philips T, Rothstein JD (2017). "Oligodendroglia: metabolic supporters of neurons". J Clin Invest. 127 (9): 3271–3280. doi:10.1172/JCI90610. PMC 5669561. PMID 28862639.
- Baumann and Pham-Dinh, 2001
- Szuchet S, Nielsen JA, Lovas G, Domowicz MS, de Velasco JM, Maric D, Hudson LD (2011). "The genetic signature of perineuronal oligodendrocytes reveals their unique phenotype". Eur J Neurosci. 34 (12): 1906–22. doi:10.1111/j.1460-9568.2011.07922.x. PMC 4286392. PMID 22132705.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Kinney, HC; Back, SA (September 1998). "Human oligodendroglial development: relationship to periventricular leukomalacia". Seminars in Pediatric Neurology. 5 (3): 180–9. doi:10.1016/s1071-9091(98)80033-8. PMID 9777676.
- Káradóttir et al., 2007
- Tkachev D, Mimmack ML, Ryan MM, et al. (September 2003). "Oligodendrocyte dysfunction in schizophrenia and bipolar disorder". Lancet. 362 (9386): 798–805. doi:10.1016/S0140-6736(03)14289-4. PMID 13678875. S2CID 7511585.
- "Chemotherapy-induced Damage to the CNS as a Precursor Cell Disease" Archived 2011-12-27 at the Wayback Machine by Dr. Mark D. Noble, University of Rochester
- Han, R; Yang, Y. M.; Dietrich, J; Luebke, A; Mayer-Pröschel, M; Noble, M (2008). "Systemic 5-fluorouracil treatment causes a syndrome of delayed myelin destruction in the central nervous system". Journal of Biology. 7 (4): 12. doi:10.1186/jbiol69. PMC 2397490. PMID 18430259.
- Fodder, Katherine; de Silva, Rohan; Warner, Thomas T.; Bettencourt, Conceição (2023-06-29). "The contribution of DNA methylation to the (dys)function of oligodendroglia in neurodegeneration". Acta Neuropathologica Communications. 11 (1): 106. doi:10.1186/s40478-023-01607-9. ISSN 2051-5960. PMC 10311741.
- Bibliography
- Raine, C.S. (1991). Oligodendrocytes and central nervous system myelin. In Textbook of Neuropathology, second edition, R.L. Davis and D.M. Robertson, eds. (Baltimore, Maryland: Williams and Wilkins), pp. 115–141.
- Káradóttir, R.; D. Attwell (14 April 2007). "Neurotransmitter receptors in the life and death of oligodendrocytes". Neuroscience. 145 (4): 1426–1438. doi:10.1016/j.neuroscience.2006.08.070. PMC 2173944. PMID 17049173.
- Gonzalez-Perez, O, B; Romero-Rodriguez, R; Soriano-Navarro, M; Garcia-Verdugo, JM; Alvarez-Buylla, A (2009). "Epidermal growth factor induces the progeny of subventricular zone type B cells to migrate and differentiate into oligodendrocytes". Stem Cells. 27 (8): 2032–43. doi:10.1002/stem.119. PMC 3346259. PMID 19544429.
- Vallstedt, A; Klos JM; Ericson F (6 January 2005). "Multiple dorsoventral origins of oligodendrocyte generation in the spinal cord and hindbrain". Neuron. 1. 45 (1): 55–67. doi:10.1016/j.neuron.2004.12.026. hdl:10616/40454. PMID 15629702. S2CID 7971750.
- Thomas, JL; Spassky N; Perez Villegas EM; Olivier C; Cobos I; Goujet-Zalc C; Martínez S; Zalc B. (15 February 2000). "Spatiotemporal development of oligodendrocytes in the embryonic brain". Journal of Neuroscience Research. 59 (4): 471–476. doi:10.1002/(SICI)1097-4547(20000215)59:4<471::AID-JNR1>3.0.CO;2-3. PMID 10679785. S2CID 42473842.
- Richardson, WD; Kessaris, N; Pringle, N (2006). "Oligodendrocyte wars". Nature Reviews Neuroscience. 1. 7 (1): 11–18. doi:10.1038/nrn1826. PMC 6328010. PMID 16371946.