Concerted reaction
In chemistry, a concerted reaction is a chemical reaction in which all bond breaking and bond making occurs in a single step. Reactive intermediates or other unstable high energy intermediates are not involved.[1][2] Concerted reaction rates tend not to depend on solvent polarity ruling out large buildup of charge in the transition state. The reaction is said to progress through a concerted mechanism as all bonds are formed and broken in concert. Pericyclic reactions, the SN2 reaction, and some rearrangements - such as the Claisen rearrangement - are concerted reactions.
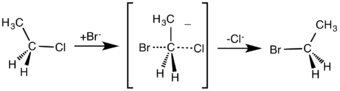
The rate of the SN2 reaction is second order overall due to the reaction being bimolecular (i.e. there are two molecular species involved in the rate-determining step). The reaction does not have any intermediate steps, only a transition state. This means that all the bond making and bond breaking takes place in a single step. In order for the reaction to occur both molecules must be situated correctly.
References
- Carey, Francis A.; Sundberg, Richard J.; (1984). Advanced Organic Chemistry Part A Structure and Mechanisms (2nd ed.). New York N.Y.: Plenum Press. ISBN 0-306-41198-9.
- "IUPAC Gold Book - concerted reaction". IUPAC. 24 February 2014. doi:10.1351/goldbook.CT07011. Retrieved 12 April 2014.
{{cite journal}}: Cite journal requires|journal=(help)