Glyptodon
Glyptodon (from Greek for "grooved or carved tooth": γλυπτός "sculptured" and ὀδοντ-, ὀδούς "tooth")[1] is a genus of glyptodontine (an extinct group of large, herbivorous armadillos) in the family Chlamyphoridae (a family of South American armadillos) that lived from the Pliocene, around 3.2 million years ago,[2] to the early Holocene, around 11,000 years ago, in Brazil, Uruguay, Paraguay, Bolivia, Peru, Argentina, and Colombia. It is one of, if not the, best known genus of glyptodont. Glyptodon has a long and storied past, being the first named extinct cingulate and the type genus of the subfamily Glyptodontinae. Fossils of Glyptodon have been recorded as early as 1814 from Pleistocene aged deposits from Uruguay, though many were incorrectly referred to the ground sloth Megatherium by early paleontologists.
| Glyptodon | |
|---|---|
 | |
| Skeleton of G. clavipes at the Naturhistorisches Museum, Vienna | |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Phylum: | Chordata |
| Class: | Mammalia |
| Order: | Cingulata |
| Family: | Chlamyphoridae |
| Subfamily: | †Glyptodontinae |
| Genus: | †Glyptodon Owen, 1839 |
| Type species | |
| †Glyptodon clavipes Owen, 1839 | |
| Other Species | |
| |
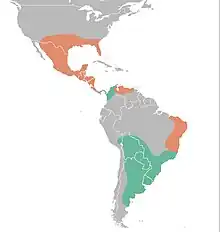 | |
| Distribution of Glyptodon (green) compared to Glyptotherium's (orange). | |
| Synonyms | |
|
Genus synonymy
Synonyms of G. clavipes
Synonyms of G. reticulatus
Dubious species
| |
The type species, G. clavipes, was described in 1839 by notable British paleontologist Sir Richard Owen. Later in the 19th century, dozens of complete skeletons were unearthed from localities and described by paleontologists such as Florentino Ameghino and Hermann Burmeister. During this era, many species of Glyptodon were dubbed, some of them based on fragmentary or isolated remains. Fossils from North America were also assigned to Glyptodon, but all of them have since been placed in the closely related genus Glyptotherium. It was not until the later end of the 1900s and 21st century that full review of the genus came about, restricting Glyptodon to just five species under one genus.
Glyptodonts were typically large, quadrapedral (four-legged), herbivorous armadillos with armored carapaces (top shell) that were made of hundreds of interconnected osteoderms (structures in dermis composed of bone). Other pieces of armor covered the tails and skull roofs, the skull being tall with hypsodont (high-crowned) teeth. As for the postcranial anatomy, pelves fused to the carapace, an amalgamate vertebral column, short limbs, and small digits are found in glyptodontines. Glyptodon reached up to 2 meters (6.56 feet) long and 400 kilograms (880 pounds) in weight, making it one of the largest glyptodontines but not as large as its close relative Glyptotherium or Doedicurus, the largest known glyptodont. Glyptodon is morphologically and phylogenetically most similar to Glyptotherium, however they differ in several ways. Glyptodon is larger on average, with an elongated carapace, a relatively shorter tail, and a robust zygoma, or cheek bone.
Glyptodonts existed for millions of years, though Glyptodon itself was one its last surviving members. Glyptodon was one of many South American megafauna, with many native groups such as notoungulates and ground sloths reaching immense sizes. Glyptodon had a mixed diet of grasses and other plants, instead living at the edge forests and grasslands where the shrubbery was lower. Glyptodon had a wide muzzle, an adaptation for bulk feeding. The armor could have protected the animal from predators, of which many coexisted with Glyptodon, including the "saber-tooth cat" Smilodon, the large canid Protocyon, and the giant bear Arctotherium.
History
Confusion with Megatherium

The history and taxonomy of Glyptodon is storied and convoluted, as it involved confusion with other genera and dubious species, as well as a lack of detailed data. The first recorded discovery of Glyptodon was as early as 1814 when Uruguayan priest, scientist, soldier, and later politician Dámaso Antonio Larrañaga (1771-1848) wrote about the discovery of several unusual fossils in his Diario de Historia Natural, which included his descriptions of many new species of ants, birds, mammals, and even one of the first figures of the extinct Megatherium, a genus of giant ground sloth that was named in 1796 by French scientist Georges Cuvier (1769-1832).[3][4] This was the first recorded discovery of a glyptodontine or fossil cingulate.[4] The unusual fossils consisted of a femur, carapace fragments, and a caudal tube (an armored tail covering found in glyptodontines) that he collected from the Pleistocene aged (ca. 2.5-0.011 mya) strata on the banks of the Solís Grande Creek, Uruguay.[4][5] Larrañaga identified the fossils as those of Dasypus (Megatherium), believing that Megatherium was a subgenus of Dasypus based on the incorrect referral of glyptodontine osteoderms to Megatherium years earlier by Spanish scientist Juan Bautista Bru de Ramón, which misled other scientists to believe that glyptodontine fossils were actually those of armored megatheres.[6][4]
Larrañaga wrote to French scientist Auguste Saint Hilaire about the discovery, and the letter was reproduced by Cuvier in 1823 in the second volume of his landmark book Recherches sur les ossemens fossiles.[7] Larrañaga also noted that similar fossils had been found in "analogous strata near Lake Merrim, on the frontier of the Portuguese colonies (southern Brazil)."[7][6] These fossils were also likely those of glyptodontines, possibly the closely related Hoplophorus.[8] The armored Megatherium hypothesis was further supported later in 1827 when portions of a Glyptodon carapace, as well as a partial femur and some caudal armor, were found by a Prussian traveler to Montevideo, Uruguay named Mr. Sellow, who sent the carapace to Berlin where it was described by Professor von Weiss, who referred it to Megatherium.[9] The femur and caudal armor were recovered from the Quegnay in northern Uruguay, while the carapace had been found in the Arapey River.[9][5] Weiss and other paleontologists noted that the osteoderms closely resembled those of armadillos like Dasypus, but Cuvier's hypothesis was popularized based on the incorrect referral of glyptodontine osteoderms Megatherium.[6][7]
Another work on the armored Megatherium hypothesis was published in 1833 by Berlin scientist E. D'Alton, who described more of the material sent by Sellow, including portions of the limbs, manus, and shoulder girdle. D'Alton recognized the great similarities of the fossils to Dasypus and speculated that it was a giant armadillo, contrary to the notion that they were from Megatherium. Despite this, D'Alton did not erect a new name for the fossils and instead wrote that additional material was necessary to distinguish it from other armadillos. D'Alton did not mention Megatherium or its osteoderms in the paper, but he implied that all of the "Megatherium armor" was instead from his armadillo. This hypothesis was supported by Laurillard in 1836, who mentioned that a plaster cast of a large armadillo carapace represented a distinct taxon from Megatherium and that the armor referred to the sloth was instead from an armadillo.[5]
1837 saw the naming of the first glyptodontine, Hoplophorus euphractus, when Danish paleontologist Peter Wilhelm Lund published a series of memoirs on the fossils of Lagoa Santa in Brazil, dating to the Pleistocene.[10][8] The fossils included osteoderms comparable to those described earlier by Larrañaga, as well as teeth, skull fragments, limb bones, and other elements.[5][8] After 1837, several new genera and species of glyptodontines were named in quick succession by European paleontologists: Chlamydotherium based on Sellow's carapace and Orycterotherium based on Sellow's femur were named by German scientist H. G. Bronn 1838,[11] Pachypus by Eduard D'Alton in 1839 based on Sellow's 1833 material,[5] Neothoracophorus (originally Thoracophorus but the name was preoccupied by a beetle) in 1889 by Argentine paleontologist Florentino Ameghino[12] based on isolated osteoderms now identified as those of a juvenile Glyptodon from Patagonia,[13] and Lepitherium in 1839 by Geoffroy Saint-Hilaire based on Sellow's osteoderms.[14][15] Saint-Hillaire considered the osteoderms found by Sellow to not even be mammal, but instead of a relative of Teleosaurus, a crocodile-like reptile known from Jurassic deposits in France.[15][14]
Richard Owen and referred species
In 1838, British diplomat Sir Woodbine Parish (1796–1882) was sent an isolated molariform and a letter about the discovery of several large fossils from the Matanza River in Buenos Aires, Argentina that dated to the Pleistocene.[16][17] Parish later collected several more fossils from localities in Las Averias and Villanueva; the latter preserved the most complete skeleton which included a mandible fragment, partial limbs, and unguals of a single individual. They were deposited in Parish's collection at the Royal College of Surgeons in the United Kingdom that year. Some of these fossils were cast at the Natural History Museum, London, but the original fossils were destroyed after German aerial bombing raids hit the college during World War II from 1940 to 1941.[13][17] Glyptodon was named by Richard Owen (1804-1892), one of the most influential British naturalists of the Victorian Era, writing a chapter about the animal and publishing a reconstruction of its skeleton in the book Buenos Ayres, and the provinces of the Rio de La Plata: their present state, trade, and debt in 1839.[18][19] Within this book, Owen erroneously believed they were all from the same specimen, the name Glyptodon ("grooved tooth") based on the anatomy of the molariform.[19][17] A later study found the molariform to actually be from another glyptodontine, Panochthus, and the Villanueva individual was designated the lectotype by Robert Hoffstetter in 1955.[20] The Las Averias individual consists of a carapace that was only mentioned in Owen's description, but was used in later reconstructions of the animal and has since been lost. An issue with the lectotype of G. clavipes is that the material is undiagnostic and indistinguishable from other Glyptodon species and even Glyptotherium, making it dubious.
Cuadrelli et al (2018) designated the species a species inquirenda due to this issue and commented that more analyses are necessary.[17] In 1860, Signor Maximo Terrero collected a partial skeleton, including a skull and carapace, of G. clavipes from the River Salado in southern Buenos Aires and dated to the Pleistocene. These fossils were also sent to the Royal College of Surgeons, where they were described in detail by British paleontologist Thomas Henry Huxley (1825–1895) in 1865 during a comprehensive review of the taxon.[5] This skeleton was also destroyed during WWII, but Huxley published several illustrations that presented great amounts of new information on the taxon.[5][21]
Later in 1845, many more fossils found by Parish from Pleistocene layers in Argentina were named as new species of Glyptodon by Owen: G. ornatus, G. reticulatus, G. tuberculatus, and G. clavicaudatus in 1847. Of these additional species, only G. reticulatus is still considered a valid species of Glyptodon as G. ornatus was reassigned to the genus Neosclerocalyptus,[22] G. tuberculatus to Panochthus,[23] and G. clavicaudatus to Doedicurus.[24] G. reticulatus was named on the basis of several carapace fragments that had also been recovered from the Matanza River, but they lack detailed locality information and the fossils too were destroyed during WWII. The fragments were cast by the NHMUK as well, being used to diagnose the species.[25]
Other paleontologists also started erecting names for Glyptodon species after the 1840s, but many of them are now seen as dubious, species inquirenda, or synonymous with previously named species.[26][17] Par L. Nodot described a new genus and species of glyptodontine in 1857, Schistopleurum typus, on the basis of a caudal tube found in the Pampas of Argentina, but it has since been synonymized with G. reticulatus.[27] Another species now seen as valid, G. munizi, was described in 1881 by Argentine paleontologist Florentino Ameghino (1853–1911) on the basis of several osteoderms found in the Ensenadan of Arroyo del Medio, San Nicolás, Argentina.[28][29] For many years the taxon was only known from the fragmentary holotype, but skull and complete carapace material of the species was later described in detail in 2006 that cemented its validity.[28][17] German zoologist Hermann Burmeister described several Glyptodon fossils in the 1860s, many of them he named as new species of Glyptodon itself or the synonym Schistopleurum, all of which are now synonyms of Glyptodon and its species.[30][17] In 1908, Florentino Ameghino named another species of Glyptodon, G. chapalmalensis, based on a carapace fragment that he had collected from the Atlantic Coast of Buenos Aires Province that dated to the Chapadmalalan. In 1932, A. Castellanos made a new genus for G. chapalmalensis, Paraglyptodon, which later included another species, P. uquiensis, that was based on more complete specimens that had been collected from Uquía, Argentina between 1909 and 1912.[31][32] The former species is dubious, but likely not Glyptodon based on its age.[33] P. uquiensis has been synonymized with Glyptodon and is possibly a valid species, though further analysis is necessary to settle its status.[25][17]

Reassessment and Glyptotherium
In the 1950s, Argentine paleontologist Alfredo Castellanos (1893-1975) erected new generic names for several species of Glyptodon, the first being Glyptocoileus and second of these being Glyptopedius in 1953 that was made for the species G. elongatus that had been named by Robert Burmeister in 1866 on the basis of a single carapace,[30] though its validity is disputed.[26][17] Castellanos also referred the species G. reticulatus to the genus, but this unsupported.[17] Yet another genus was erected in 1976 named Heteroglyptodon genuarioi by F. L. Roselli based on an incomplete skeleton that had been collected from the Pleistocene aged Libertad Formation in Nueva Palmira, Uruguay,[34][35] but it has since been found to be an indeterminate specimen of Glyptodon.[35] Several Glyptodon fossils from Pleistocene deposits in Colombia were described in 2012, extending the known range of the genus north greatly.[36]
Another Glyptodon species was described in 2020 called G. jatunkhirkhi by several authors led by Argentine zoologist Francisco Cuadrelli on the basis of an individual preserving a nearly complete carapace, several caudal rings, and a pelvis that had been collected from Yamparaez, 24 kilometres (15 mi) southeast of the Bolivian city of Sucre. The strata they were found in was made up of fluvial, sandy sediments that dated to the Late Pleistocene from elevations as high as 4,100–2,500 metres (13,500–8,200 ft) above sea level.[26] Several additional paratypes were referred to the species from other Late Pleistocene sites in Eastern Cordillera, Bolivia including a nearly complete skull and several osteoderms.[26] In a phylogenetic analysis conducted by Cuadrelli et al., 2020, G. jatunkhirki was recovered as the most basal Glyptodon species, despite being the same age as the more derived species G. clavipes. Reassesment of Glyptodon species began in the late 20th and early 21st centuries, with various hypotheses developing on the number of valid species. Numbers varied, with some authors considering up to 4 species valid, while phylogenetic analyses in 2018 and 2020 only found the species G. reticulatus, G. munizi, and G. jatunkhirkhi definitively valid; G. clavipes and G. uquiensis as species inquirendas.[17] However a 2016 review of G. uquiensis determined that G. uquiensis was actually a juvenile specimen of Glyptodon, though the species could not be determined.[33]
Fossils from North America were also described and referred to Glyptodon starting in 1875, when civil engineers J. N. Cuatáparo and Santiago Ramírez collected a skull, nearly complete carapace, and associated postcranial skeleton of a glyptodontine from a drainage canal near Tequixquiac, Mexico, the fossils coming from the Rancholabrean stage of the Pleistocene.[37][25] These fossils were the first found of glyptodontines in North America and were named as a new species of Glyptodon, G. mexicanum, but the fossils have since been lost and the species was synonymized with Glyptotherium cylindricum.[25][38] Several other North American glyptodontine species were named throughout the late 19th-early 20th century, typically based on fragmentary osteoderms. All North American and Central American fossils of glyptodontines have since been referred to the closely related genus Glyptotherium, which was named in 1903 by American paleontologist Henry Fairfield Osborn.[39]
Taxonomy
Glyptodon is the type genus of Glyptodontinae, an extinct subfamily of large, heavily armored armadillos that first evolved in the Late Eocene (ca. 33.5 mya) and went extinct in the Early Holocene during the late Pleistocene extinctions (ca. 7,000 years ago).[26][40] Owen recognized that Glyptodon was an edentate, but did not recognize it as being a part of a new subfamily as there were no other recognized glyptodontines in 1839.[19] The family Glyptodontidae was not named until 1869 by John Edward Gray, who included the genera Glyptodon, Panochthus, and Hoplophorus within the group and believed that it was diagnosed by an immovable carapace that was fused to the pelvis.[41] However, Hermann Burmeister proposed the name Biloricata for the family, believing that glyptodontines possessed a ventral plastron (bottom shell) and could pull their heads inside their carapaces like turtles. This name lost all use and his theory has not been supported.[30][23] The internal phylogenetics of Glyptodontidae was analyzed in greater detail by Florentino Ameghino during his descriptions of earlier members of the clade, which proposed that Glyptodon was descended from earlier genera.[12][28]
Glyptodontinae was classified in its own family or even superfamily until in 2016, when ancient DNA was extracted from the carapace of a 12,000 year old Doedicurus specimen, and a nearly complete mitochondrial genome was reconstructed (76x coverage). Comparisons with those of modern armadillos revealed that glyptodonts diverged from tolypeutine and chlamyphorine armadillos approximately 34 million years ago in the late Eocene.[42][40] This prompted moving them from their own family, Glyptodontidae, to the subfamily Glyptodontinae within the extant Chlamyphoridae.[40] Based on this and the fossil record, glyptodonts would have evolved their characteristic shape and large size (gigantism) quite rapidly, possibly in response to the cooling, drying climate and expansion of open savannas.[42] Chylamyphoridae is a group in the order Cingulata, which includes all extant armadillos in addition to other fossil groups like Pachyarmatheriidae and Pampatheridae. Cingulata is itself within the basal mammal group Xenarthra, which includes an array of American mammal groups like Vermilingua (anteaters) and Folivora (sloths and ground sloths) in the order Pilosa. The following phylogenetic analysis was conducted by Frédéric Delsuc and colleagues in 2016 and represents the phylogeny of Cingulata using ancient DNA from Doedicurus to determine the position of it and other Glyptodonts:[42][40]
| Cingulata |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

The internal phylogeny of Glyptodontinae is convoluted and in flux, with many species and families erected based on fragmentary or undiagnostic material that lacks comprehensive review.[43][8] Glyptodontinae's tribes were long-considered subfamilies before the 2016 analysis.[44] One tribe, Glyptodontini (typically labeled Glyptodontinae) is a group of younger, larger glyptodontines that evolved in the Middle Miocene (ca. 13 mya) with Boreostemma,[45] but split into two genera, Glyptodon in the south and Glyptotherium in the north,[36] though Glyptotherium also lived in some areas of South America like Venezuela and eastern Brazil.[46][47][36] Glyptotherium and Glyptodon lived during the same intervals and are nearly identical to Glyptodon in many aspects, so much so that the first fossils of Glyptotherium to be described were misidentified as those of Glyptodon.[48][49][25] Glyptodontini is distinguishable from other groups for example in that it has large, conical tubercular osteoderms absent or only present on the caudal (tailward) notch on the posterior end of the carapace and different ornamentation of the armor on the carapace than the tail.[25] Glyptodontini is often recovered as more basal to most other glyptodontines like Doedicurus, Hoplophorus, and Panochthus.[50][26]
Below is the phylogenetic analysis conducted by Cuadrelli et al., 2020 of Glyptodontinae, with Glyptodontidae as a family instead of subfamily, that focuses on advanced glyptodonts:[26]
| Chlamyphoridae |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Description

Like the extant armadillos and all other glyptodontines, Glyptodon had a large, bony carapace that covered much of its torso, as well as smaller cephalic armor covering the roof of its head, akin to that in turtles. The carapace was composed of hundreds of small, hexagonal osteoderms (armored structures made of bone), with Glyptodon carapaces preserving a total of 1,800 osteoderms each. The anatomy of different Glyptodon species varies greatly, mostly in the species G. jatunkhirkhi which is more similar to Glyptotherium in certain aspects.[26]
In the axial skeleton, glyptodontines had strongly fused vertebrae and pelves completely connected to the carapace, traits convergently evolved in turtles.[5][38] The large tails of glyptodontines likely served as a counterbalance to the rest of the body and Glyptodon's caudal armor ended in a blunt tube that was composed of two concentric tubes fused together, in contrast to those of mace-tailed glyptodontines like Neosclerocalyptus and Doedicurus.[38] Glyptodon had graviportal (weight-bearing), short limbs that are very similar to those in other glyptodontines, being indistinguishable from those of some other taxa.[25] The digits of Glyptotherium are very stout and adapted for weight-bearing, though some preserve large claw sheaths that had an intermediate morphology between claws and hooves.[38]
During the Pleistocene, the diversity of glyptodontines diminished but body size increased, with the largest known glyptodont, Doedicurus, evolving in the Pleistocene.[51][52] Glyptodon sizes vary between species and individuals. G. clavipes, the type species, was estimated to weigh 2,000 kilograms (4,400 lb),[53][54] G. reticulatus weighed a mere 401 kilograms (884 lb) to 862 kilograms (1,900 lb),[55] and G. munizi weighed 1,150 kilograms (2,540 lb).[56] A partial skeleton of G. clavipes measured 3.5 metres (11 ft) with a carapace length of 1.7 metres (5.6 ft),[4][21] while the carapaces of other species like G. munizi and G. reticulatus measured 2.2 metres (7.2 ft) and 2.19 metres (7.2 ft) long respectively.[25]
Skull, mandible, and dentition

Glyptodont dentition contains entirely hypsodont molariforms, which have one of the most extreme examples of hypsodonty known from terrestrial mammals.[57] The dentition is typical of other armadillos, but is fluted on each side by deep grooves. The anterior teeth were compressed, while the posterior teeth were cylindrical.[58] Glyptodont skulls have several unique features; the maxilla and palatine are enlarged vertically to make space for the molariforms, while the braincase is brachycephalic, short and flat.[59] In Glyptodon and many other glyptodontines, the roof of the skull was covered by a shield composed of polygonal, irregular osteoderms that were variable in size and ankylosed together to form a robust cephalic shield that had a smoothly convex exterior surface without ornamentation.[28] Each osteoderm has a rugose and slightly convex dorsal surface, with ornamentation pattern defined by a central figure, slightly elevated and surrounded by an area without peripheral figures or foramina. Sutures separating osteoderms are well marked, as in Panochthus.[28][60] Other Pleistocene glyptodontines are known by complete/sub-complete skulls, allowing for comparisons to Glyptodon.[13][43] Glyptotherium's zygoma are narrow, slender, almost parallel, and close to the sagittal plane in frontal view; in Glyptodon, this structure is broader, robust, divergent rather than parallel and more laterally placed.[51][25]
The nasal passage was reduced with heavy muscle attachments for some unknown purpose. Some have speculated that the muscle attachments were for a proboscis, or trunk, much like that of a tapir or elephant. The lower jaws were very deep and helped support massive chewing muscles to help chew coarse fibrous plants. Some paleontologists have proposed that Glyptodon and some glyptodontines also had a proboscis or large snout similar to those in proboscideans and tapirs,[61] but few have accepted this hypothesis.[43][62] Another suggestion, made by A.E. Zurita and colleagues, is that the large nasal sinuses could be correlated with the cold arid climate of Pleistocene South America.[62][63] A distinctive bar of bone projects downwards on the cheek, extending over the lower jaw, perhaps providing an anchor for powerful snout muscles. In turn, the infraorbital foramina are narrow and not visible in anterior view in Glyptotherium, but in Glyptodon they are broad and clearly visible in anterior view. In lateral view, the dorso-ventral height between the skull roof and the palatal plane in Glyptodon decreases anteriorly, contrary to Glyptotherium; the nasal tip is in a lower plane with respect to the zygomatic arch in Glyptodon, but in Glyptotherium is higher than the zygomatic arch plane. The 1st molariform (molaiform is abbreviated as mf#) of Glyptodon is distinctly trilobate (three-lobed) both lingually and labially, nearly as trilobate as the mf2; on the contrary, Glyptotherium shows a very low trilobation of mf1, which is elliptical in cross-section, the mf2 is weakly trilobate, and the mf3 is trilobate. In both genera, the mf4 to mf8 are fully trilobate and serially identical.[25] These traits separate the two genera.[25] Within the genus Glyptodon this trait varies as well, with G. reticulatus having triloby to a greater degree than G. munizi.[17]

The mandibles of Glyptotherium and Glyptodon are very similar, but Glyptotherium's mandible is smaller by about 10% in total size. The angle between the occlusal plane (part of the jaw where upper and lower teeth contact) and the anterior margin of the ascending ramus is approximately 60 in Glyptotherium, while it is 65° in Glyptodon. The ventral margin of the horizontal ramus is more concave in Glyptodon than in Glyptotherium. The symphysis area is extended greatly in Glyptotherium antero-posteriorily compared to Glyptodon. The mf1 is ellipsoidal in Glyptotherium and the mf2 is "submolariform", while in Glyptodon both teeth are trilobate.[25]
Vertebrae and pelvis
)_BHL39943960.jpg.webp)
Glyptodon has 7 cervical vertebrae, of which the first 3 cervicals were fused together while the rest of the cervicals were free except for the 7th.[5] The 7th cervical and the first 2 dorsal vertebrae were fused together into a trivertebral, a broad, flat bone with very small spinous processes (projections from a vertebra) and large articular surfaces that held ribs.[5] All of the other 13 vertebrae in the dorsal column were fused into one long continuous tunnel that is not seen in mammals outside of glyptodontines, some of these vertebrae were so tightly fused that the segments of them cannot be discerned. The centra of these vertebrae were curved, thin bony plates that created a cylinder to support the carapace and the shape of the animal.[5] Spinous processes in these vertebrae are also heavily reduced, with some being only a thin blade of bone ankylosed with other vertebrae.[5] Sacral vertebrae in Glyptodon are also fused and 13 in number, which preserve very unusual oval-shaped, thin, and slightly concave ends on the centra.[5] The pelves are also unusual, as they preserve giant ilia and are fused to the rest of the skeleton.[5]
Carapace and osteoderms


Glyptodon's osteoderms were attached by synotoses (bony connections) and were found in double or triple rows on the front and sides of the carapace's edges, as well as in the tail armor and cephalic shield. The carapace's osteoderms were conical with a rounded point, while the ones on the tail were just conical. The sulci between these raised structures were deep and wide with parallel lines.[64] The carapace of Glyptodon was strongly elongated compared to those of Boreostemma and Glyptotherium, with the carapace being relatively 65% longer than the former and 14% than the latter. In Glyptodon, the top-bottom height of the carapace represents 60% of its total length, whereas in Glyptotherium it is taller at circa 70%. The antero-posterior dorsal profile of the carapace was convex and its posterior half was higher than the anterior. The apex of the carapace was slightly displaced posteriorly in most Glyptodon species, while in Glyptotherium and Glyptodon jatunkhirkhi it was at the center of the midline. The carapace of most species of Glyptodon is arched subtly, while Glyptotherium and Glyptodon jatunkhirkhi's has a very arched back and convex pre-iliac and concave post-iliac, giving it a saddle-like overhang over the tail. Glyptodon osteoderms in the antero-lateral regions of the carapace are strongly ankylosed, giving them little flexibility, while in Glyptotherium they are less ankylosed and more flexible.[25] The osteoderms of the caudal aperture (large conical osteoderms that protect the base of the tail) are more conical in Glyptodon and more rounded in Glyptotherium, though in the latter the anatomy of the caudal aperture osteoderms varies by sex while in Glyptodon it varies by age.[25][65] The caudal aperture is more vertically oriented in the latter genus, while in Glyptotheirum it is angled posteriorily.[25] Although frequently used to differentiate the two taxa, Glyptodon and Glyptotherium have similar osteoderm morphologies that differ only in several areas. Both genera have tall, thick osteoderms compared to those of many other glyptodontines such as Hoplophorus and Neosclerocalyptus. Glyptodon sometimes preserves a "rosette" pattern, where the osteoderm's central figure is surrounded by a row of peripheral figures, while other specimens lack them completely. G. reticulatus varies from a complete rosette pattern to a reticular surface, which has convex central and peripheral figures.[65][25] Glyptotherium however always preserves rosettes.[65][25] The central and radial sulci are deeper and broader in Glyptodon (ca. 4–6 mm) than in Glyptotherium (ca. 1–2.4 mm). The osteoderms in Glyptodon and Glyptotherium have 5-11 peripheral figures, rugose exposed surfaces, and heights up to 47 millimetres (1.9 in).[25]
Osteoderms on the ventral side of the body were first mentioned by paleontologist Hermann Burmeister in 1866, postulating that there was a ventral plastron like in turtles based on evidence of small armor in the dermis.[30] This hypothesis has since been disproven, but in the early 2000s, the presence of osteoderms on Glyptodon's face, hind legs, and underside was confirmed in several species. The fossils with these characteristics were from the Pleistocene, evolving in younger species like G. reticulatus . These small to medium-sized ossicles were actually embedded in the dermis and did not connect in a pattern.[64]
Tail

Glyptodon had very primitive tail anatomy for a glyptodont, possessing eight or nine mobile caudal rings of fused, large, conical osteoderms. These enclosed the base of the tail, which terminated in a short caudal tube composed of two fused caudal rings. Caudal rings were composed of two or three rows of pentagonal osteoderms that transitioned from flat, slightly convex in the posterior rings to conical tubercles by the third caudal ring. The more posterior the rings were, the larger they were, with the exception of the 2nd ring which was the largest and 1st complete ring in the series, creating a cone-shaped tail. The distal scutes are larger, and their free margins are rounded producing a fan-like shape. Most of the osteoderms of the distal row (some individuals preserving up to 12) bear prominent conical outlines, in stark contrast to more advanced glyptodontines like Doedicurus and Panochthus, which had completely fused tails that formed an inflexible mace or club.[66] The caudal tube at the distalmost end of the tail is cylinder-shaped with smaller conical osteoderms and is stubbier proportionally in Glyptodon. In Glyptotherium, this caudal tube represents ca. 20% of the total length of the caudal armor, whereas in Glyptodon, this structure represents 13% of the total length.[25] In Glyptodon, the caudal armor length represents circa 30-40% of the carapace's total length in contrast to Glyptotherium, where this value is greater at around 50%.[25] For example, in specimen MCA 2015 of Glyptodon reticulatus, the terminal tube measured only 73.23 millimetres (2.883 in) long in comparison to Glyptotherium texanum specimen UMMP 34 826's 210 millimetres (8.3 in) long tube.[25]
Paleobiology
Digging abilities
.jpg.webp)
Many armadillo species have digging capabilities, with large claws adapted for scraping dirt in order to make burrows or forage for food underground.[67][68] Much of armadillo diets consist of insects and other invertebrates that live underground,[69] in contrast to the herbivorous diets of Glyptodon and related genera.[70] Being a large armadillo, Glyptodon's fossorial capabilities have been researched on several occasions. Owen (1841) opposed this idea,[16] though pushback came from Nodot (1856) and Sénéchal (1865) who believed digging was possible for the genus.[27][71] However, the evolution of a rigid carapace as opposed to a flexible one in extant armadillos as well as a weakly developed deltoid crest on the humerus (upper arm bone) provided evidence against fossorial hypotheses. The elbow had a great range of movement, as with digging cingulates, but this is more likely to be due to size adaptations.[55][72]
Endocranial anatomy
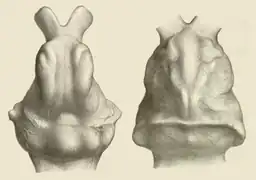
Several complete skulls of Glyptodon enable the endocranial anatomy to be analyzed, as well as compared to other well-preserved taxa like Doedicurus and Panochthus. The brain cavities of the larger glyptodontines Glyptodon, Doedicurus, and Panochthus had a braincase volume of 213 to 234 cubic centimeters (7.2 to 7.9 U.S. fl oz). The encephalization quotient of these taxa are 0.12 to 0.4, lower than most modern armadillos (0.44-1.06) and corresponds to those of pampatheres. The brain of the glyptodontines had an extensive olfactory bulb that took up between 4.8 and 9.7% of the entire brain, while around two thirds of it were occupied by the cerebrum and the rest by the cerebellum. Overall, this is akin to that of other armadillos, but in the latter the cerebrum is smaller relative to the cerebellum and the braincase's total volume. Deviating from the armadillos with their wide olfactory bulb, glyptodontines and pampatheres have elongated and triangular olfactory systems. Several other neuroanatomical characteristics differ between glyptodontines and armadillos, such as the presence of a pronounced sulcus praesylvianus.[73][74]
In general, living cingulates have smaller brains than anteaters and sloths for reasons unknown. Several theories have been made as to why, such as a shorter rearing phase of offspring, dedication of resources to the development of the carapace, and other biological and functional handicaps. Members of Cingulata also tend to have extremely low metabolisms, causing less energy flow to the development of the brain's neurons. The pattern of large bodies bearing adequate protection and a reduction of intelligence is found in several other groups such as ankylosaurs and stegosaurs, two types of armored dinosaur. However, the carapace itself is considered as a restrictive functional component as it prohibited much neck movement and forced a reduced brain size. This reduction thus resulted in weight loss in the skull, which had a great affect on the skulls of large-headed glyptodontines like Glyptodon.[73][74]
Feeding and diet

Two main groups of glyptodontines can be distinguished by their feeding habits: narrow-muzzled Miocene propalaehoplophorids and wide-muzzled post-Miocene glyptodontines.[75] The propalaehoplophorids were selective feeders, while the post-Miocene glyptodontines were bulk feeders (obtain nutrients by consuming an entire plant). However, because of their body form and fusion of the cervical vertebrae glyptodontines would have needed to forage near the ground. Their craniomandibular joint limited their jaw to side-to-side movement.[76] Glyptodon's jaws had large ridges of osteodentine which could effectively be used to grind food particles before shearing and pushing them via the constant motion of the mandible.[76] They had a well-developed snout musculature, along with a mobile neck region that helped them secure food.[77] The hyoid shows a robust design that suggests Glyptodon had a large and robust tongue, which may have aided in food intake and processing.[78][79]
Like most other xenarthrans, glyptodontines had lower energy requirements than contemporary mammal groups.[80] The stomachs of glyptodontids are mysterious due to being entirely herbivorous, in contrast to modern, omnivorous armadillos which have simple stomachs instead of the chambered ones of sloths.[81] This in conjugation with the proposed idea that aquatic grazing may have caused the isotopes strongly associated with herbivory observed in Glyptodon fossils.[81] However, aquatic grazing in Glyptodon is little supported[82] though more backing for this hypothesis has been found in the related Glyptotherium.[83][84][38] A carbon isotopic analysis of Glyptodon bones by França et al (2015) found that it consumed a variety of both C3 plants and C4 grasses at lower latitudes while it ate exclusively C3 grasses at higher ones, implying an ecological shift based on the climate. A 2012 analysis of isotopes supports this, but the isotopic results are not backed by morphological evidence.[76] The isotopic conclusion would place Glyptodon as a mixed browser in most environments, similar to some other glyptodontines.[85][86] The 2012 paper also noted that Glyptodon may have had a more flexible diet than previously imagined,[87] with a mix of slightly wooded and slightly open habitats as implied by the consumption of C3 and C4 material.[86][56] The C4 plants include groups like Poaceae, Cyperaceae, Asteraceae, and Amaranthaceae based on palynological evidence, meaning that Glyptodon likely ate C4 flowering plants in addition to C3 grasses.[88][85] A mesowear analysis supported their conclusion, however, finding that mixed-feeding causing blunt wear that suggests a more abrasion-dominated diet. This is similar to that of Neosclerocalyptus, but in contrast to Hoplophorus which had sharper wear ends. Neosclerocalyptus favored more open environments despite this, as found by isotopic studies.[43][56] The mesowear angles of Glyptodon were noted to possess a bimodal distribution, implying a difference between populations, sexes, or species in diet.[70]
Intraspecific combat
.jpg.webp)
Glyptodonts are believed to have taken part in intraspecific fighting. It was presumed that since the tail of Glyptodon was very flexible and had rings of bony plates, it was used as a weapon in fights. Although its tail could be used for defense against predators, evidence suggests that the tail of Glyptodon was primarily for attacks on its own kind. A G. reticulatus fossil displays damage done on the surface of its carapace. A study based on this specimen calculated that Glyptodon tails would have been able to generate enough force to break the carapace of another Glyptodon. This suggests that they likely fought each other to settle territorial or mating disputes through the use of their tails, much like male-to-male fighting among deer using their antlers. .[89]
Ontogeny
In 2009, a partial skeleton of a prenatal individual of Glyptodon was described that had been found inside of the pelvic region of a carapace of an adult.[90] The skeleton had been collected from the Pleistocene-aged deposits in the Tarija Valley of Bolivia and included a partial skull, partial mandible, and fragments from the scapulae and femora. The skeleton is the only known prenatal specimen of a glyptodontine and is one of the most complete specimens of an immature Glyptodon known, though dozens of isolated osteoderms from juveniles are known.[90] The preserved skull measures only 51 mm long, but still bears many characteristics of Glyptodon such as a subtriangular naris, a lateral margin on the naris that forms an acute angle of 30-degrees, oval infraorbital foramina, and several other traits.[90] However, the mandible differs in that the ascending ramus is at a 90-degree angle in contrast to the 60-70 degree angles preserved in adults. Interestingly, this mandibular morphology is alike to that in some specimens of Glyptotherium cylindricum.[60][90]
In the osteoderms of juvenile Glyptodon reticulatus, the central figures are larger than the peripheral osteoderms. These central figures are planar, sometimes even concave, and elevated compared to the peripherals. The peripherals in younger individuals are also less distinct and bear weakly marked or absent furrows (grooves that separate osteoderms). On the other hand, peripherals and central figures of adults are similarly sized, distinct, and of similar heights.[17][25]
Posture
Several interpretations of glyptodontine posture have been made,[91] starting with those by Richard Owen in 1841 using comparative anatomy.[16] Owen theorized that the phalanges were weight-bearing due to their short and broad physiology, in addition to evidence provided in the postcranial skeleton.[16] It was also proposed that an upright posture was possible for Glyptodon, first by Sénéchal (1865) who stated that the tail could be an equilibrium for the front half of the body as well as a method of supporting the legs.[71] Linear measurements were later taken which provided insight into this hypothesis, finding that bipedalism would be possible.[92][93] The patellar articulation with the femur suggests rotation of the lower leg during knee extension and potentially even knee-locking were feasible.[94]
Sexual dimorphism and group behavior
No evidence of sexual dimorphism in Glyptodon has been described, but it has been observed in the close relative Glyptotherium based on fossils found in Pliocene deposits in Arizona. In the genus, the caudal aperture of males and females differ in that the marginal osteoderms of males are much more conical and convex than those of females. Even in the carapaces of newborn Glyptotherium, the marginal osteoderms are either conical or flat, which enables their sex to be determined.[65] No direct evidence of glyptodontine group behavior has been described, though some localities preserving juveniles, subadults, and adults of Glyptotherium together are known.[65][51] Living armadillos are loners and only come together during mating season, with the number of offspring varying between one and even twelve babies depending on the species.[95]
Distribution and paleoecology
Glyptodon is one of the most common Pleistocene glyptodontines with a large range from the lowland Pampas to the towering Andean Mountains of Peru and Bolivia, some fossils found at elevations reaching over 4,100 metres (13,500 ft) above sea level.[96][97][26] Only G. munizi is found in the early-middle Pleistocene, whereas other species are younger.[28][26] G. reticulatus is specifically noted to be known from 60ka to as recent as 7ka possibly, though confirmed records only extend to 11 ka.[98] The genus had a generalist diet, which allowed it to fill niches in areas that were inaccessible by grazing genera, with G. reticulatus representing up to 90% of the glyptodontine fossils in the Tarija Valley of Bolivia.[13] However, in regions such as the Pampas, Mesopotamia, and Uruguay, an array of glyptodontines are known.[98][17] Further evidence of Glyptodon's adaptability is found in the Pampas, which were semihumid and temperate from 30,000 to 11,000 ka, alternating between the rainy and dry seasons, over a large area consisting mostly of grasslands dotted with forests and mixed shrubbery.[99][100][17] Temperatures in this region were lower than the present, with an estimated mean annual temperature 4.2 °C (39.6 °F) in the Pampas compared to 16.4 °C (61.5 °F) in Buenos Aires today. The Pampas specifically was a mix of semi-arid Patagonian and tropical Brazilian climates during the middle Pleistocene before the expansion of the drier climates.[101] This is in stark contrast to the Bermejo Formation of Formosa Province, Argentina where the climate and fauna suggest a more arid environment with fewer grasslands.[102][103] G. jatunkhirkhi specifically is known only from Andean climate of Eastern Cordillera in Bolivia, causing it to evolve to be smaller in size than lowland species due to less support for larger masses.[26][97] G. jatunkhirkhi is not the only example of this in Xenarthra, with species of Panochthus and Pleurolestodon evolving to be smaller in size in mountainous regions.[97][26]

During the Ensenadan and Marplatan, Glyptodon coexisted with a variety of mammals unique to the period such as the notoungulate Mesotherium, canid Theriodictis, and a species of the giant bear Arctotherium angustidens.[101] In areas such as Uruguay, fossils of Glyptodon have been unearthed alongside the contemporary glyptodontines Doedicurus, Neuryurus, Panochthus; armadillos Chaetophractus, Propaeopus, and Eutatus; and the insectivorous pampathere Pampatherium. As for their distant relatives the ground sloths, the humongous Megatherium is known, in addition to two species of the scelidothere Catonyx, and the mylodontid genera Mylodon and Glossotherium. Some other groups are known, including the unusual litopternans Macrauchenia and Neolicaphrium, notoungulate Toxodon, massive proboscidean Notiomastodon, and the equids Equus neogeus and Hippidion. Various artiodactyls have been recorded, including the peccaries Catagonus and Tayassu peccari, extinct deers Morenelaphus and Antifer, and two genera of llamas including Hemiauchenia and Lama. A variety of carnivorans have been recorded, such as the "saber-toothed" Smilodon, the bear Arctotherium bonariense, and the wolf-like canids Protocyon, and Dusicyon.[104][105] Rodents too have been found, such as Holochilus, Hydrochoerus (capybara), Cavia, and Microcavia.[98] Notably, some of the youngest "terror-bird" fossils of the genus Psilopterus have been unearthed in the area.[106]
Material previously assigned to Glyptodon in northeast Brazil has been reassigned to Glyptotherium, restricting the Brazilian distribution of Glyptodon to the southern provinces. However, two osteoderms with characteristics similar to those of Glyptodon have been found in Sergipe state in the northeast, suggesting that both genera occurred in this region during the Pleistocene.[47] Glyptodon's northernmost locality comes from Pleistocene deposits in central Colombia,[36] though many specimens formerly attributed to the genus come from the bordering country of Venezuela.[51]
Predation and relationship with humans
Glyptodon coexisted with a variety of large predators including the cat Smilodon, jaguar, and canid Protocyon.[107][108] This belief is furthered by the discovery of fractured dorsal armor, which implies that Glyptodon had been in physical conflict with other animals.[64] However, isotope analyses of the collagen from Glyptodon and other mammals of the Pampas region by Bocherens et al. (2015) discovered little evidence to support the idea of predators feeding on Glyptodon.[107] Instead, it was found that Glyptodon as well as herbivorous mammals living in denser forests made up a smaller portion of carnivore diets, whereas open grazers such as Lestodon and Macrauchenia were consumed more often.[107] Furthermore, the appearance of secondary armor in the dermis of Glyptodon coincides with the arrival of North American predators in South America during the Great American Interchange.[2] For this reason, it was hypothesized that the osteoderms developed as a defensive/offensive mechanism to combat the new arrivals of the area.[64]
Smilodon may have occasionally preyed upon glyptodontines, based on a skull of Glyptotherium texanum which bears the distinctive elliptical puncture marks that best match those of the machairodont cat, indicating that the predator successfully bit into the skull through the armored cephalic shield.[109] The Glyptotherium in question was a juvenile, with a still-developing head shield, making it far more vulnerable to the cat's attack.[110] Although originally theorized by George Brandes to be possible in 1900,[111] Smilodon canines could not pierce the thick carapace osteoderms of glyptodontines.[112] Brandes imagined that the evolution of thick glyptodontine armor and long machairodont canines was an example of coevolution,[111] but Birger Bohlin argued in 1940 that the teeth were far too fragile to do damage against glyptodontine armor.[112][64]

The coexistence of early hunter-gatherer humans and glyptodontines in South America was first hypothesized in 1881 based on fossil discoveries from the Pampas,[113] and many fossil discoveries from the Late Pleistocene to Early Holocene have been unearthed since that exhibit human predation on glyptodontines. No fossils of Glyptodon preserving direct interactions have been unearthed, but it did inhabit this region alongside humans. At the site of Pay Paso 1, an archaeological site in northwestern Uruguay preserving human-made spear points and other signs of culture were found associated with fossils of Glyptodon and the horse Equus. These were used for radiocarbon dating using collagen, supposedly dating to around 9,000 to 9,500 BP but these dates cannot be verified.[114][25] During this period, a wide array of Xenarthrans inhabited the Pampas were hunted by humans, with evidence demonstrating that the small (300–450 kg, 660–990 lb) glyptodontine Neosclerocalyptus,[115] the armadillo Eutatus, and the gigantic (2 ton) glyptodontine Doedicurus, the largest glyptodontine known, were hunted.[116] The only other records of human predation from outside the Pampas area a partial carapace, which was eviscerated by humans, and several skulls preserving signs that they were dispatched by human tools. All were found in Venezuela.[117][51] The discoveries there showed the first signs of human hunting on the skulls of glyptodontines.[118] Hunters may have used the shells of dead animals as shelters in inclement weather.[119][120]
Extinction
Some evidence suggests that humans drove glyptodontines to extinction.[121] Evidence from the Campo Laborde and La Moderna archaeological sites in the Argentine Pampas suggest that Glyptodon's relatives Doedicurus and Panochthus survived until the Early Holocene, coexisting with humans for a minimum of 4,000 years.[122] This overlap provides support for models showing that the South American Pleistocene extinctions resulted from a combination of climatic change and anthropogenic causes.[122] These sites have been interpreted as ones used for butchering megafauna (Megatherium and Doedicurus); however, some of the chronology has been problematic and controversial, due to poor preservation of the collagen used for dating.[122] The extinction rates in South America during the Quaternary Extinction Event were the highest out of any continent, with all endemic animals weighing over 100 kilograms (220 lb) going extinct by the middle Holocene.[4] This supports the idea of human hunting as a drive for the extinction of Glyptodon, as the arrival of humans around 16,000 years BP to such a formerly isolated continent may have caused extinction rates to become higher.[118][4][123]
The extinction of Glyptodon notably coincides with the end of the Antarctic Cold Reversal period in which, for 1,700 years, temperatures dropped before spiking after ending at 12.7 ka.[124][125] Many climatic fluctuations occurred during the late Pleistocene between humid and dry cycles, with Glyptodon preferring drier climates. Following the Antarctic Cold Reversal, temperatures rose and the climate became more consistently humid, which then led C3 grasses to become increasingly replaced by C4 grasses and southern beech trees. These changes led vulnerable, grazing-specialized forms like glyptodontines, toxodonts, and some ground sloths to become extinct.[126][127] Around 11.5 ka, temperatures peaked before again dropping, resulting in the extinction of several different genera of mammals including some megafauna. Glyptodon along with genera such as Glossotherium and Morenelaphus were wiped out, though several other groups lived for several thousand years after.[44][98]
References
- "glyptodon". Oxford English Dictionary (1st ed.). Oxford University Press. 1933.
- Zurita, Alfredo E.; Carlini, Alfredo A.; Gillette, David; Sánchez, Rodolfo (2011-03-01). "Late Pliocene Glyptodontinae (Xenarthra, Cingulata, Glyptodontidae) of South and North America: Morphology and paleobiogeographical implications in the GABI". Journal of South American Earth Sciences. 31 (2): 178–185. Bibcode:2011JSAES..31..178Z. doi:10.1016/j.jsames.2011.02.001. ISSN 0895-9811.
- Cuvier, G. (1796). Notice sur le squelette d'une très-grande espèce de quadrupède inconnue jusqu'à présent, trouvé au Paraguay, et déposé au cabinet d'histoire naturelle de Madrid. de l'imprimerie du Magasin encyclopédique, rue Honoré No 94, vis-à-vis le passage Roch.
- Fariña, Richard A.; Vizcaíno, Sergio F.; De Iuliis, Gerry (22 May 2013). Megafauna: Giant Beasts of Pleistocene South America. Indiana University Press. ISBN 978-0-253-00719-3. OCLC 779244424.
- Huxley, Thomas Henry (1865-01-01). "II. On the osteology of the genus glyptodon". Philosophical Transactions of the Royal Society of London. 155: 31–70. doi:10.1098/rstl.1865.0002. S2CID 111224208.
- Owen, Professor (1855). "On the Megatherium (Megatherium Americanum, Cuvier and Blumenbach). Part II.--Vertebrae of the Trunk". Philosophical Transactions of the Royal Society of London. 145: 359–388. ISSN 0261-0523. JSTOR 108525.
- Cuvier, Georges (1823). Recherches sur les ossemens fossiles (in French). chez G. Dufour et E. d'Ocagne.
- Porpino, Kleberson de O.; Fernicola, Juan C.; Bergqvist, Lílian P. (2010-05-18). "Revisiting the intertropical Brazilian species Hoplophorus euphractus (Cingulata, Glyptodontoidea) and the phylogenetic affinities of Hoplophorus". Journal of Vertebrate Paleontology. 30 (3): 911–927. Bibcode:2010JVPal..30..911P. doi:10.1080/02724631003765735. ISSN 0272-4634. S2CID 83888299.
- Weiss, C. S. (1827). Über das südliche ende des gebirgszuges von Brasilien in der provinz S. Pedro do Sul und der Banda oriental oder dem staate von Monte Video. Verlag nicht ermittelbar.
- Lund, P. W. (1837). Blik paa Brasiliens dyreverden foÈr sidste jordomvaeltning. Popp.
- Bronn, H. G. (1858). Untersuchungen über die Entwickelungs-Gesetze der organischen Welt während der Bildungs-Zeit unserer Erd-Oberfläche. E. Schweizerbart.
- Ameghino, F. (1889). Contribucion al conocimiento de los mamiferos fosiles de la República Argentina: Obra escrita bajo los auspicios de la Academia nacional de ciencias de la República Argentina para ser presentada á la Exposicion universal de Paris de 1889 (Vol. 6). PE Coni é hijos.
- Zurita, A. E., Miño-Boilini, Á. R., Soibelzon, E., Carlini, A. A., & Paredes Rios, F. (2009). The diversity of Glyptodontidae (Xenarthra, Cingulata) in the Tarjia Valley (Bolivia): Systematic, biostratigraphic and paleobiogeographic aspects of a particular assemblage.(With 3 figures and 1 table). Neues Jahrbuch fur Geologie und Palaontologie-Abhandlungen, 251(2), 225.
- Podgorny, Irina (2013). "Fossil dealers, the practices of comparative anatomy and British diplomacy in Latin America, 1820–1840". The British Journal for the History of Science. 46 (4): 647–674. doi:10.1017/S0007087412000702. ISSN 0007-0874. S2CID 147258227.
- Saint-Hilaire, E. G. (1831). Recherches sur de grands sauriens: trouvés à l'état fossile vers les confins maritimes de la basse normandie, attribués d'abord au crocodile, puis déterminés sous les noms de téléosaurus et sténéosaurus. Firmin Didot.
- Owen, R. (1841). VI.—Description of a Tooth and Part of the Skeleton of the Glyptodon clavipes, a large Quadruped of the Edentate Order, to which belongs the Tesselated Bony Armour described and figured by Mr. Clift in the former Volume of the Transactions of the Geological Society; with a consideration of the question whether the Megatherium possessed an analogous Dermal Armour. Transactions of the Geological Society of London, 6(1), 81-106.
- Cuadrelli, Francisco; Zurita, Alfredo E.; Toriño, Pablo; Miño-Boilini, Ángel R.; Rodríguez-Bualó, Santiago; Perea, Daniel; Acuña Suárez, Gabriel E. (2018-09-03). "Late Pleistocene Glyptodontinae (Mammalia, Xenarthra, Glyptodontidae) from southern South America: a comprehensive review". Journal of Vertebrate Paleontology. 38 (5): e1525390. Bibcode:2018JVPal..38E5390C. doi:10.1080/02724634.2018.1525390. ISSN 0272-4634. S2CID 92335544.
- Parish, W. (1852). Buenos Ayres, and the provinces of the Rio de La Plata: their present state, trade, and debt. Murray.
- Owen, R. (1839). Note on the Glyptodon. Buenos Aires and the Provinces of the Rio de La Plata, 1-178.
- Hoffstetter, R. (1955). Sur le genotype de Glyptodon Owen. Bulletin du Muséum National d’Historie Naturelle, 27(5), 408-413.
- Huxley, Thomas Henry (1862-01-01). "Description of a New Specimen of Glyptodon, Recently Acquired by the Royal College of Surgeons of England". Proceedings of the Royal Society of London. 12: 316–326. doi:10.1098/rspl.1862.0071. JSTOR 112260.
- Paula Couto, C. D. (1957). Sôbre um gliptodonte do Brasil. Boletim Divisão de Geologia e Mineralogia, 165, 1-37.
- Burmeister, G. (1866). Lista de los mamíferos fósiles del terreno diluviano. In Anales del Museo Público de Buenos Aires (Vol. 1, No. 3, pp. 121-232).
- Burmeister, G. Burmeister 1870–1874. Monografia de los glyptodontes en el Museo Público de Buenos Aires. Anales del Museo Público de Buenos Aires, 2, 1-412.
- Zurita, Alfredo Eduardo; Gillette, David D.; Cuadrelli, Francisco; Carlini, Alfredo Armando (2018-06-01). "A tale of two clades: Comparative study of Glyptodon Owen and Glyptotherium Osborn (Xenarthra, Cingulata, Glyptodontidae)". Geobios. 51 (3): 247–258. Bibcode:2018Geobi..51..247Z. doi:10.1016/j.geobios.2018.04.004. ISSN 0016-6995. S2CID 134450624.
- Cuadrelli, Francisco; Zurita, Alfredo E.; Toriño, Pablo; Miño-Boilini, Ángel R.; Perea, Daniel; Luna, Carlos A.; Gillette, David D.; Medina, Omar (2020-09-16). "A new species of glyptodontine (Mammalia, Xenarthra, Glyptodontidae) from the Quaternary of the Eastern Cordillera, Bolivia: phylogeny and palaeobiogeography". Journal of Systematic Palaeontology. 18 (18): 1543–1566. doi:10.1080/14772019.2020.1784300. ISSN 1477-2019. S2CID 221064742.
- Nodot, L. (1856). Description d'un nouveau genre d'édenté fossile renfermant plusieurs espèces voisines du glyptodon: suivie d'une nouvelle méthode de classification applicable à toute l'histoire naturelle, et spécialement à ces animaux, avec un atlas de douze planches lithographiées; ouvrage publié par l'Academie des sciences, arts et belles-lettres de Dijon et l'atlas avec le concours du Conseil municipal de même ville. Imprimerie Loireau-Feuchot.
- Soibelzon, Esteban; Zurita, Alfredo Eduardo; Carlini, Alfredo A. (2006). "Glyptodon munizi Ameghino (Mammalia, Cingulata, Glyptodontidae): redescripción y anatomía". Ameghiniana. 43 (2): 377–384. ISSN 0002-7014.
- Ameghino, F. 1882. Catálogo de las colecciones de Antropología prehistórica y paleontología de Florentino Ameghino, Partido de Mercedes. En: Catálogo de la Sección de la Provincia de Buenos Aires (República Argentina). Exposición Continental Sudamericana. Anexo A: 35-42.
- Burmeister, H. (1866). XXXIX.—On Glyptodon and its allies. Annals and Magazine of Natural History, 18(106), 299-304.
- Castellanos, A. (1932). Nuevos géneros de gliptodontes en relación con su filogenia.
- Castellanos, A. (1953). Descripción de restos de" Paraglyptodon uquiensis" n. sp. de Uquía (Senador Pérez) de Jujuy (No. 32). la Provincia.
- Cruz, Laura E.; Fernicola, Juan C.; Taglioretti, Matias; Toledo, Nestor (2016-03-01). "A reassessment of the taxonomic status of Paraglyptodon Castellanos, 1932 (Mammalia, Cingulata, Glyptodontia)". Journal of South American Earth Sciences. 66: 32–40. Bibcode:2016JSAES..66...32C. doi:10.1016/j.jsames.2015.11.012. ISSN 0895-9811.
- Jiménez Zamudio, Rafael (1988-06-30). "Contribución al estudio del plural de la declinación temática latina". Emerita. 56 (1): 121–126. doi:10.3989/emerita.1988.v56.i1.612. ISSN 1988-8384.
- Zurita, Alfredo Eduardo; Oliveira, Edison Vicente; Toriño, Pablo; Rodriguez-Bualó, Santiago Martín; Scillato-Yané, Gustavo Juan; Luna, Carlos; Krapovickas, Jerónimo (2011-01-01). "On the taxonomic status of some Glyptodontidae (Mammalia, Xenarthra, Cingulata) from the Pleistocene of South America". Annales de Paléontologie. 97 (1): 63–83. Bibcode:2011AnPal..97...63Z. doi:10.1016/j.annpal.2011.07.003. hdl:11336/43130. ISSN 0753-3969.
- Zurita, Alfredo Eduardo; Miño Boilini, Ángel Ramón; Francia, Analia; Arenas Mosqueras, José E. (2012). "The Pleistocene Glyptodontidae Gray, 1869 (Xenarthra: Cingulata) of Colombia and some considerations about the South American Glyptodontinae". Rev. Bras. Paleontol. 15 (3): 273–280. doi:10.4072/rbp.2012.3.04. ISSN 1519-7530.
- Cuatáparo, J. N., & Ramírez, S. (1875). Descripción de un mamífero fósil de especie desconocida perteneciente al género" Glyptodon": encontrado entre las capas post-terciarias de Tequisquiac, en el Distrito de Zumpango. F. Diaz de Leon.
- Gillette, David D.; Ray, Clayton E. (1981). "Glyptodonts of North America" (PDF). Smithsonian Contributions to Paleobiology (40): 1–255. doi:10.5479/si.00810266.40.1.
- Osborn, Henry Fairfield (1903). Glyptotherium Texanum, a New Glyptodont, from the Lower Pleistocene of Texas. order of the Trustees, American Museum of Natural History.
- Delsuc, F.; Gibb, G.C.; Kuch, M.; Billet, G.; Hautier, L.; Southon, J.; Rouillard, J.-M.; Fernicola, J.C.; Vizcaíno, S.F.; MacPhee, R.D.E.; Poinar, H.N. (2016). "The phylogenetic affinities of the extinct glyptodonts". Current Biology. 26 (4): R155–R156. doi:10.1016/j.cub.2016.01.039. PMID 26906483.
- Gray, J. E. (1869). Catalogue of carnivorous, pachydermatous, and edentate Mammalia in the British Museum. order of the Trustees.
- Mitchell, K.J.; Scanferla, A.; Soibelzon, E.; Bonini, R.; Ochoa, J.; Cooper, A. (2016). "Ancient DNA from the extinct South American giant glyptodont Doedicurus sp. (Xenarthra: Glyptodontidae) reveals that glyptodonts evolved from Eocene armadillos". Molecular Ecology. 25 (14): 3499–3508. doi:10.1111/mec.13695. PMID 27158910. S2CID 3720645.
- Zurita, A.E.; Scarano, A.C.; Carlini, A.A.; Scillato-Yané, G.J.; Soibelzon, E. (2011-04-04). "Neosclerocalyptus spp. (Cingulata: Glyptodontidae: Hoplophorini): cranial morphology and palaeoenvironments along the changing Quaternary". Journal of Natural History. 45 (15–16): 893–914. doi:10.1080/00222933.2010.536917. ISSN 0022-2933. S2CID 85146482.
- Mitchell, Kieren J.; Scanferla, Agustin; Soibelzon, Esteban; Bonini, Ricardo; Ochoa, Javier; Cooper, Alan (2016). "Ancient DNA from the extinct South American giant glyptodont Doedicurus sp. (Xenarthra: Glyptodontidae) reveals that glyptodonts evolved from Eocene armadillos". Molecular Ecology. 25 (14): 3499–3508. doi:10.1111/mec.13695. PMID 27158910. S2CID 3720645.
- Zurita, Alfredo E.; González Ruiz, Laureano R.; Gómez-Cruz, Arley J.; Arenas-Mosquera, Jose E. (2013-05-01). "The most complete known Neogene Glyptodontidae (Mammalia, Xenarthra, Cingulata) from northern South America: taxonomic, paleobiogeographic, and phylogenetic implications". Journal of Vertebrate Paleontology. 33 (3): 696–708. Bibcode:2013JVPal..33..696Z. doi:10.1080/02724634.2013.726677. ISSN 0272-4634. S2CID 86099989.
- Oliveira, É. V., Porpino, K. D., & Baretto, A. (2010). On the presence of Glyptotherium in the Late Pleistocene of Northeastern Brazil, and the status of "Glyptodon" and "Chlamydotherium". Paleobiogeographic implications. Neues Jahrbuch fur Geologie und Palaontologie-Abhandlungen, 258(3), 353.
- Dantas, M. A. T.; França, L. M.; Cozzuol, M. A.; Rincón, A. D. (2013). "About the occurrence of Glyptodon sp. in the Brazilian intertropical region". Quaternary International. 305: 206–208. Bibcode:2013QuInt.305..206D. doi:10.1016/j.quaint.2011.06.024.
- Hay, O. P. (1923). The Pleistocene of North America and Its Vertebrated Animals from the States East of the Mississippi River and from the Canadian Provinces East of Longitude 95> O (No. 22). Carnegie institution of Washington.
- Cope, E. D. (1889). the edentata of North America. The American Naturalist, 23(272), 657-664.
- Porpino, Kleberson de O.; Fernicola, Juan C.; Cruz, Laura Edith; Bergqvist, Lílian P. (2014-07-29). "The intertropical Brazilian species of Panochthus (Xenarthra, Cingulata, Glyptodontoidea): a reappraisal of their taxonomy and phylogenetic affinities". Journal of Vertebrate Paleontology. 34 (5): 1165–1179. Bibcode:2014JVPal..34.1165P. doi:10.1080/02724634.2014.863203. hdl:11336/29660. ISSN 0272-4634. S2CID 86577502.
- Carlini, Alfredo A.; Carrillo-Briceño, Jorge D.; Jaimes, Arturo; Aguilera, Orangel; Zurita, Alfredo E.; Iriarte, José; Sánchez-Villagra, Marcelo R. (2022-06-16). "Damaged glyptodontid skulls from Late Pleistocene sites of northwestern Venezuela: evidence of hunting by humans?". Swiss Journal of Palaeontology. 141 (1): 11. doi:10.1186/s13358-022-00253-3. ISSN 1664-2384. S2CID 249681104.
- Defler, Thomas (2019), Defler, Thomas (ed.), "The Xenarthrans: Armadillos, Glyptodonts, Anteaters, and Sloths", History of Terrestrial Mammals in South America: How South American Mammalian Fauna Changed from the Mesozoic to Recent Times, Topics in Geobiology, Cham: Springer International Publishing, vol. 42, pp. 117–138, doi:10.1007/978-3-319-98449-0_6, ISBN 978-3-319-98449-0, S2CID 92413294, retrieved 2022-07-19
- Fariña, R. A., Vizcaíno, S. F., & Bargo, M. S. (1998). Body mass estimations in Lujanian (late Pleistocene-early Holocene of South America) mammal megafauna. Mastozoología Neotropical, 5(2), 87-108.
- Delsuc, F.; Gibb, G.C.; Kuch, M.; Billet, G.; Hautier, L.; Southon, J.; Rouillard, J.-M.; Fernicola, J. C.; Vizcaíno, S. F.; MacPhee, R. D.E.; Poinar, H. N. (2016-02-22). "The phylogenetic affinities of the extinct glyptodonts" (PDF). Current Biology. 26 (4): R155–R156. doi:10.1016/j.cub.2016.01.039. PMID 26906483.
- Vizcaíno, Sergio F.; Blanco, R. Ernesto; Bender, J. Benjamín; Milne, Nick (2011). "Proportions and function of the limbs of glyptodonts: Glyptodont limbs". Lethaia. 44 (1): 93–101. doi:10.1111/j.1502-3931.2010.00228.x.
- Vizcaíno, Sergio F.; Cassini, Guillermo H.; Fernicola, Juan C.; Bargo, M. Susana (2011). "Evaluating Habitats and Feeding Habits Through Ecomorphological Features in Glyptodonts (Mammalia, Xenarthra)". Ameghiniana. 48 (3): 305–319. doi:10.5710/AMGH.v48i3(364). ISSN 0002-7014. S2CID 85793531.
- Gillette & Ray 1981, p. 200.
- Flower, W.H. (1871). "Professor Flower's Hunterian Lectures On The Teeth and Allied Organs In The Mammalia". The British Medical Journal.
- Gillette & Ray 1981, p. 58.
- Carlini, Alfredo A.; Zurita, Alfredo E.; Aguilera, Orangel A. (2008). "North American Glyptodontines (Xenarthra, Mammalia) in the Upper Pleistocene of northern South America". Paläontologische Zeitschrift. 82 (2): 125. doi:10.1007/BF02988404. S2CID 128528271.
- Gillette & Ray 1981, p. 202.
- Fernicola, Juan Carlos; Néstor Toledo; M. Susana Bargo; Sergio F. Vizcaíno (October 2012). "A neomorphic ossification of the nasal cartilages and the structure of paranasal sinus system of the glyptodont Neosclerocalyptus Paula Couto 1957 (Mammalia, Xenarthra)". Palaeontologia Electronica. 15 (3): 1–22. doi:10.26879/333.
- Gillette, David D. (2010). "Glyptodonts in Arizona a saga of supercontinents, sea-floor spreading, savannas, and sabertooth cats". Arizona Geological Survey. Retrieved 25 March 2014.
- Zurita, A. E.; Soibelzon, L. H.; Soibelzon, E.; Gasparini, G. M.; Cenizo, M. M.; Arzani, H. (2010). "Accessory protection structures in Glyptodon Owen (Xenarthra, Cingulata, Glyptodontidae)". Annales de Paléontologie. 96 (1): 1–11. Bibcode:2010AnPal..96....1Z. doi:10.1016/j.annpal.2010.01.001. hdl:10915/5356.
- Gillette, David D.; Carranza-Castañeda, Óscar; White, Richard S.; Morgan, Gary S.; Thrasher, Larry C.; McCord, Robert; McCullough, Gavin (2016-06-01). "Ontogeny and Sexual Dimorphism of Glyptotherium texanum (Xenarthra, Cingulata) from the Pliocene and Pleistocene (Blancan and Irvingtonian NALMA) of Arizona, New Mexico, and Mexico". Journal of Mammalian Evolution. 23 (2): 133–154. doi:10.1007/s10914-015-9309-6. ISSN 1573-7055. S2CID 254699205.
- Arbour, Victoria M.; Zanno, Lindsay E. (2020). "Tail Weaponry in Ankylosaurs and Glyptodonts: An Example of a Rare but Strongly Convergent Phenotype". The Anatomical Record. 303 (4): 988–998. doi:10.1002/ar.24093. ISSN 1932-8486. PMID 30835954. S2CID 73488683.
- Vizcaíno, S. F., Fariña, R. A., & Mazzetta, G. V. (1999). Ulnar dimensions and fossoriality in armadillos. Acta Theriologica, 44.
- Carter, T. S., & Encarnaçao, C. D. (1983). Characteristics and use of burrows by four species of armadillos in Brazil. Journal of Mammalogy, 64(1), 103-108.
- Vizcaíno, Sergio F.; Fariña, Richard A. (2007-03-29). "Diet and locomotion of the armadillo Peltephilus: a new view". Lethaia. 30 (1): 79–86. doi:10.1111/j.1502-3931.1997.tb00446.x.
- Saarinen, Juha; Karme, Aleksis (2017-06-15). "Tooth wear and diets of extant and fossil xenarthrans (Mammalia, Xenarthra) – Applying a new mesowear approach". Palaeogeography, Palaeoclimatology, Palaeoecology. 476: 42–54. Bibcode:2017PPP...476...42S. doi:10.1016/j.palaeo.2017.03.027. ISSN 0031-0182.
- Sénéchal, D. L. (1865). Notice sur l'armure ou le dermato-squelette et le système dentaire du Glyptodon clavipes, et particularités biologiques de cet animal, déduites d'après l'étude de ses restes fossiles. Balitout, Questroy et Cie.
- Amson, Eli; Nyakatura, John A. (2018). "The Postcranial Musculoskeletal System of Xenarthrans: Insights from over Two Centuries of Research and Future Directions". Journal of Mammalian Evolution. 25 (4): 459–484. doi:10.1007/s10914-017-9408-7. ISSN 1064-7554. S2CID 254693335.
- Tambusso, P Sebastián; Fariña, Richard A. (2015-09-03). "Digital cranial endocast of Pseudoplohophorus absolutus (Xenarthra, Cingulata) and its systematic and evolutionary implications". Journal of Vertebrate Paleontology. 35 (5): e967853. Bibcode:2015JVPal..35E7853T. doi:10.1080/02724634.2015.967853. ISSN 0272-4634. S2CID 86537473.
- Sebastián Tambusso, P.; Fariña, Richard A. (2015). "Digital endocranial cast of Pampatherium humboldtii (Xenarthra, Cingulata) from the Late Pleistocene of Uruguay". Swiss Journal of Palaeontology. 134 (1): 109–116. doi:10.1007/s13358-015-0070-5. ISSN 1664-2384. S2CID 86652394.
- "Bargo M. S. Vizcaíno S. F. — Paleobiology of Pleistocene ground sloths (Xenarthra, Tardigrada) : biomechanics, morphogeometry and ecomorphology applied to the masticatory apparatus. Ameghiniana". ResearchGate. Retrieved 2015-10-30.
- Fariña, R.A. (2001). "Carved Teeth and Strange Jaws: How Glyptodonts Masticated" (PDF). Acta Palaeontologica Polonica.
- Gillette, R. (21 December 1981). "Glyptodonts of North America" (PDF). Smithsonian Publications. Retrieved 2015-10-29.
- Pérez, Leandro M.; Toledo, Néstor; De Iuliis, Gerardo; Bargo, M. Susana; Vizcaíno, Sergio F. (2010). "Morphology and function of the hyoid apparatus of fossil xenarthrans (mammalia)". Journal of Morphology. 271 (9): 1119–1133. doi:10.1002/jmor.10859. PMID 20730924. S2CID 8106788.
- Zamorano, M., Scillato-Yané, G. J., Soibelzon, E., Soibelzon, L. H., Bonini, R., & Rodriguez, S. (2018). Hyoid apparatus of Panochthus sp.(Xenarthra; Glyptodontidae) from the Late Pleistocene of the Pampean region (Argentina). Comparative description and muscle reconstruction. Neues Jahrbuch für Geologie und Paläontologie Abhandlungen, 288, 205-219.
- Vizcaíno, Sergio F.; Cassini, Guillermo H.; Fernicola, Juan C.; Bargo, M. Susana (2011). "Evaluating Habitats and Feeding Habits Through Ecomorphological Features in Glyptodonts (Mammalia, Xenarthra)". Ameghiniana: 305–319. doi:10.5710/AMGH.v48i3(364). S2CID 85793531. Retrieved 2015-10-29.
- Bocherens, Hervé; Cotte, Martin; Bonini, Ricardo A.; Straccia, Pablo; Scian, Daniel; Soibelzon, Leopoldo; Prevosti, Francisco J. (2017). "Isotopic insight on paleodiet of extinct Pleistocene megafaunal Xenarthrans from Argentina". Gondwana Research. 48: 7–14. Bibcode:2017GondR..48....7B. doi:10.1016/j.gr.2017.04.003.
- Fariña, R.A., 1995. Limb bone strength and habits in large glyptodonts. Lethaia, 28: 189-196.
- Lessa, Carlos Micael Bonfim; Gomes, Verônica Santos; Cherkinsky, Alexander; Dantas, Mário André Trindade (2021-12-01). "Isotopic paleoecology (δ13C, δ18O) of two megamammals assemblages from the late pleistocene of Brazilian intertropical region". Journal of South American Earth Sciences. 112: 103576. Bibcode:2021JSAES.11203576L. doi:10.1016/j.jsames.2021.103576. ISSN 0895-9811. S2CID 244181217.
- Omena, Érica Cavalcante; Silva, Jorge Luiz Lopes da; Sial, Alcides Nóbrega; Cherkinsky, Alexander; Dantas, Mário André Trindade (2021-10-03). "Late Pleistocene meso-megaherbivores from Brazilian Intertropical Region: isotopic diet ( δ 13 C), niche differentiation, guilds and paleoenvironmental reconstruction ( δ 13 C, δ 18 O)". Historical Biology. 33 (10): 2299–2304. doi:10.1080/08912963.2020.1789977. ISSN 0891-2963. S2CID 225543776.
- França, Lucas de Melo; de Asevedo, Lidiane; Dantas, Mário André Trindade; Bocchiglieri, Adriana; Avilla, Leonardo dos Santos; Lopes, Renato Pereira; da Silva, Jorge Luíz Lopes (2015-01-01). "Review of feeding ecology data of Late Pleistocene mammalian herbivores from South America and discussions on niche differentiation". Earth-Science Reviews. 140: 158–165. Bibcode:2015ESRv..140..158F. doi:10.1016/j.earscirev.2014.10.006. ISSN 0012-8252.
- Domingo, Laura; Prado, José Luis; Alberdi, María Teresa (2012-11-08). "The effect of paleoecology and paleobiogeography on stable isotopes of Quaternary mammals from South America". Quaternary Science Reviews. 55: 103–113. Bibcode:2012QSRv...55..103D. doi:10.1016/j.quascirev.2012.08.017. ISSN 0277-3791.
- Pomi, L. H. (2008). Una nueva asociación de vertebrados fósiles de Edad Ensenadense (Plioceno tardío-Pleistoceno medio) de la provincia de Buenos Aires, Argentina. Ameghiniana, 45(2), 503-510.
- Sage, Rowan F. (2004). "The evolution of C 4 photosynthesis". New Phytologist. 161 (2): 341–370. doi:10.1111/j.1469-8137.2004.00974.x. ISSN 0028-646X. PMID 33873498.
- Alexander, R. M.; Fariña, R. A.; Vizcaíno, S. F. (May 1999). "Tail blow energy and carapace fractures in a large glyptodont (Mammalia, Xenarthra)". Zoological Journal of the Linnean Society. 126 (1): 41–49. doi:10.1111/j.1096-3642.1999.tb00606.x.
- Zurita, Alfredo Eduardo; Miño-Boilini, Angel R.; Soibelzon, Esteban; Scillato-Yané, Gustavo J.; Gasparini, Germán M.; Paredes-Ríos, Freddy (2009-09-01). "First record and description of an exceptional unborn specimen of Cingulata Glyptodontidae: Glyptodon Owen (Xenarthra)". Comptes Rendus Palevol. 8 (6): 573–578. Bibcode:2009CRPal...8..573Z. doi:10.1016/j.crpv.2009.04.003. hdl:11336/37710. ISSN 1631-0683.
- Amson, Eli; Nyakatura, John A. (2018-12-01). "The Postcranial Musculoskeletal System of Xenarthrans: Insights from over Two Centuries of Research and Future Directions". Journal of Mammalian Evolution. 25 (4): 459–484. doi:10.1007/s10914-017-9408-7. ISSN 1573-7055. S2CID 254693335.
- Fari a, R. A., & Vizcaíno, S. F. (1997). Allometry of the bones of living and extinct armadillos (Xenarthra, Dasypoda). Zeitschrift fur Saugetierkunde, 62, 65-70.
- Fariña, Richard A.; Vizcaı́no, Sergio F.; Blanco, R. Ernesto (1997-04-21). "Scaling of the Indicator of Athletic Capability in Fossil and Extant Land Tetrapods". Journal of Theoretical Biology. 185 (4): 441–446. Bibcode:1997JThBi.185..441F. doi:10.1006/jtbi.1996.0323. ISSN 0022-5193.
- Shockey, B. J. (2001). Specialized knee joints in some extinct, endemic, South American herbivores. Acta Palaeontologica Polonica, 46(2).
- Superina, M. (2000). Biologie und Haltung von Gürteltieren (Dasypodidae) (Doctoral dissertation, Uitgever niet vastgesteld).
- Pujos, François; Salas, Rodolfo (2004-08-01). "A systematic reassessment and paleogeographic review of fossil Xenarthra from Peru". Bulletin de l'Institut français d'études andines. 33 (2): 331–377. doi:10.4000/bifea.5746. ISSN 0303-7495. S2CID 130927479.
- Zurita, Alfredo Eduardo; Zamorano, Martín; Scillato-Yané, Gustavo Juan; Fidel, Sergio; Iriondo, Martín; Gillette, David D. (2017-11-17). "A new species of Panochthus Burmeister (Xenarthra, Cingulata, Glyptodontidae) from the Pleistocene of the Eastern Cordillera, Bolivia". Historical Biology. 29 (8): 1076–1088. doi:10.1080/08912963.2016.1278443. ISSN 0891-2963. S2CID 91031708.
- Ubilla, Martín; Rinderknecht, Andrés; Corona, Andrea; Perea, Daniel (2018-06-01). "Mammals in Last 30 to 7 ka Interval (Late Pleistocene-Early Holocene) in Southern Uruguay (Santa Lucía River Basin): Last Occurrences, Climate, and Biogeography". Journal of Mammalian Evolution. 25 (2): 291–300. doi:10.1007/s10914-017-9380-2. ISSN 1573-7055. S2CID 254697032.
- Blasi, A., Castiñeira Latorre, C., Del Puerto, L., Prieto, A. R., Fucks, E., De Francesco, C., ... & Young, A. (2010). Paleoambientes de la cuenca media del río Luján (Buenos Aires, Argentina) durante el último período glacial (EIO 4-2). Latin American journal of sedimentology and basin analysis, 17(2), 85-111.
- Prado, José L.; Martinez-Maza, Cayetana; Alberdi, María T. (2015-05-01). "Megafauna extinction in South America: A new chronology for the Argentine Pampas". Palaeogeography, Palaeoclimatology, Palaeoecology. 425: 41–49. Bibcode:2015PPP...425...41P. doi:10.1016/j.palaeo.2015.02.026. ISSN 0031-0182.
- Prado, José Luis; Alberdi, María Teresa; Bellinzoni, Jonathan (2021). "Pleistocene Mammals from Pampean Region (Argentina). Biostratigraphic, Biogeographic, and Environmental Implications". Quaternary. 4 (2): 15. doi:10.3390/quat4020015. ISSN 2571-550X.
- Kruck, W., Helms, F., Geyh, M. A., Suriano, J. M., Marengo, H. G., & Pereyra, F. (2011). Late pleistocene-holocene history of chaco-pampa sediments in Argentina and Paraguay. E&G Quaternary Science Journal, 60(1), 14.
- Zurita, A. E., M. Taglioretti, M. De los Reyes, C. Oliva, and F. Scaglia. 2014. First Neogene skulls of Doedicurinae (Xenarthra, Glyptodontidae): morphology and phylogenetic implications. Historical Biology 28:423–432.
- Prevosti, F.J.; Ubilla, M.; Perea, D. (2009). "Large extinct canids from the Pleistocene of Uruguay: systematic, biogeographic and paleoecological remarks". Historical Biology. 21 (1–2): 79–89. doi:10.1080/08912960903249337. hdl:11336/103575. ISSN 0891-2963. S2CID 85140125.
- Manzuetti, Aldo; Perea, Daniel; Rinderknecht, Andrés; Ubilla, Martín (2018-09-01). "New Canid Remains from Dolores Formation, late Pleistocene-early Holocene, Uruguay". Journal of Mammalian Evolution. 25 (3): 391–396. doi:10.1007/s10914-017-9387-8. ISSN 1573-7055. S2CID 254701482.
- Jones, Washington; Rinderknecht, Andrés; Alvarenga, Herculano; Montenegro, Felipe; Ubilla, Martín (2018-06-01). "The last terror birds (Aves, Phorusrhacidae): new evidence from the late Pleistocene of Uruguay". PalZ. 92 (2): 365–372. doi:10.1007/s12542-017-0388-y. ISSN 1867-6812. S2CID 134344096.
- Bocherens, Hervé; Cotte, Martin; Bonini, Ricardo; Scian, Daniel; Straccia, Pablo; Soibelzon, Leopoldo; Prevosti, Francisco J. (2016-05-01). "Paleobiology of sabretooth cat Smilodon populator in the Pampean Region (Buenos Aires Province, Argentina) around the Last Glacial Maximum: Insights from carbon and nitrogen stable isotopes in bone collagen". Palaeogeography, Palaeoclimatology, Palaeoecology. 449: 463–474. Bibcode:2016PPP...449..463B. doi:10.1016/j.palaeo.2016.02.017. ISSN 0031-0182.
- Montalvo, C. I., Zárate, M. A., Bargo, M. S., & Mehl, A. (2013). Registro faunístico y paleoambientes del Cuaternario tardío, provincia de La Pampa, Argentina. Ameghiniana, 50(6), 554-570.
- Antón, Mauricio (2013). Sabertooth. Bloomington, Indiana: University of Indiana Press. pp. 203–204. ISBN 978-0-253-01042-1.
- Gillette, D. D. (Spring 2010). "Glyptodonts in Arizona". Arizona Geology. Arizona Geological Survey. Retrieved 2018-08-17.
- Brandes, G. (1900) : Ueber eine Ursache des Aussterbens Diluvialer Säugethiere. Corrblatt d. Deutsch . Ges. f. Anthropol. Jahrg. 31. Munichen 1901.
- Bohlin, B. (1940). 8. Food habit of the machairodonts, with special regard to Smilodon.
- Vogt, C. (1881). Squelette humain associe aux glyptodontidae. Bulletin de la Societe d’Antropologie de Paris, 3(4), 693–699
- Suárez, Rafael (2015). "The Paleoamerican Occupation of the Plains of Uruguay: Technology, Adaptations, and Mobility". PaleoAmerica. 1 (1): 88–104. doi:10.1179/2055556314Z.00000000010. ISSN 2055-5563. S2CID 128985617.
- Quiñones, Sofía I.; De los Reyes, Martin; Zurita, Alfredo E.; Cuadrelli, Francisco; Miño-Boilini, Ángel R.; Poiré, Daniel G. (2020-11-01). "Neosclerocalyptus Paula Couto (Xenarthra, Glyptodontidae) in the late Pliocene-earliest Pleistocene of the Pampean region (Argentina): Its contribution to the understanding of evolutionary history of Pleistocene glyptodonts". Journal of South American Earth Sciences. 103: 102701. Bibcode:2020JSAES.10302701Q. doi:10.1016/j.jsames.2020.102701. ISSN 0895-9811. S2CID 225024450.
- Politis, Gustavo G.; Messineo, Pablo G.; Stafford, Thomas W.; Lindsey, Emily L. (2019). "Campo Laborde: A Late Pleistocene giant ground sloth kill and butchering site in the Pampas". Science Advances. 5 (3): eaau4546. Bibcode:2019SciA....5.4546P. doi:10.1126/sciadv.aau4546. ISSN 2375-2548. PMC 6402857. PMID 30854426.
- Bryan, Alan L.; Casamiquela, Rodolfo M.; Cruxent, José M.; Gruhn, Ruth; Ochsenius, Claudio (1978-06-16). "An El Jobo Mastodon Kill at Taima-taima, Venezuela". Science. 200 (4347): 1275–1277. Bibcode:1978Sci...200.1275B. doi:10.1126/science.200.4347.1275. ISSN 0036-8075. PMID 17738722. S2CID 42568763.
- Prates, Luciano; Perez, S. Ivan (2021-04-12). "Late Pleistocene South American megafaunal extinctions associated with rise of Fishtail points and human population". Nature Communications. 12 (1): 2175. Bibcode:2021NatCo..12.2175P. doi:10.1038/s41467-021-22506-4. ISSN 2041-1723. PMC 8041891. PMID 33846353.
- Fidalgo, F., et al. (1986) "Investigaciones arqueológicas en el sitio 2 de Arroyo Seco (Pdo. de Tres Arroyos, prov. de Buenos Aires, República Argentina)" In: Bryan, Alan (ed.) (1986) New evidence for the Pleistocene peopling of the Americas Peopling of the Americas Symposia Series, Center for the Study of Early Man, University of Maine, Orono, Maine, ISBN 0-912933-03-8, pp. 221–269, in Spanish
- Politis, Gustavo G. and Gutierrez, Maria A. (1998) "Gliptodontes y Cazadores-Recolectores de la Region Pampeana (Argentina)" ("Glyptodonts and Hunter-Gatherers in the Pampas Region (Argentina)") Latin American Antiquity9(2): pp.111–134 in Spanish
- Martin, P. S.; Steadman, D. W. (1999-06-30). "Prehistoric extinctions on islands and continents". In MacPhee, R. D. E (ed.). Extinctions in near time: causes, contexts and consequences. Advances in Vertebrate Paleobiology. Vol. 2. New York: Kluwer/Plenum. pp. 17–56, see p. 38. ISBN 978-0-306-46092-0. OCLC 41368299. Retrieved 2015-11-07.
- Politis, G. G.; Messineo, P. G. (November 2008). "The Campo Laborde site: New evidence for the Holocene survival of Pleistocene megafauna in the Argentine Pampas". Quaternary International. 191 (1): 98–114. Bibcode:2008QuInt.191...98P. doi:10.1016/j.quaint.2007.12.003.
- Prates, Luciano; Politis, Gustavo G.; Perez, S. Ivan (2020-07-22). "Rapid radiation of humans in South America after the last glacial maximum: A radiocarbon-based study". PLOS ONE. 15 (7): e0236023. Bibcode:2020PLoSO..1536023P. doi:10.1371/journal.pone.0236023. ISSN 1932-6203. PMC 7375534. PMID 32697794.
- Orombelli, Giuseppe; Maggi, Valter; Delmonte, Barbara (2010-06-01). "Quaternary stratigraphy and ice cores". Quaternary International. Plio-Pleistocene Correlation and Global Change. 219 (1): 55–65. Bibcode:2010QuInt.219...55O. doi:10.1016/j.quaint.2009.09.029. ISSN 1040-6182.
- Weber, M. E.; Clark, P. U.; Kuhn, G.; Timmermann, A.; Sprenk, D.; Gladstone, R.; Zhang, X.; Lohmann, G.; Menviel, L.; Chikamoto, M. O.; Friedrich, T.; Ohlwein, C. (2014). "Millennial-scale variability in Antarctic ice-sheet discharge during the last deglaciation". Nature. 510 (7503): 134–138. Bibcode:2014Natur.510..134W. doi:10.1038/nature13397. ISSN 1476-4687. PMID 24870232. S2CID 205238911.
- Villavicencio, Natalia A.; Werdelin, Lars (2018-09-01). "The Casa del Diablo cave (Puno, Peru) and the late Pleistocene demise of megafauna in the Andean Altiplano". Quaternary Science Reviews. 195: 21–31. Bibcode:2018QSRv..195...21V. doi:10.1016/j.quascirev.2018.07.013. ISSN 0277-3791. S2CID 134626837.
- Hubbe, Alex; Hubbe, Mark; Karmann, Ivo; Cruz, Francisco W.; Neves, Walter A. (2011). "Insights into Holocene megafauna survival and extinction in southeastern Brazil from new AMS 14C dates". Quaternary Research. 79 (2): 152–157. doi:10.1016/j.yqres.2012.11.009. ISSN 0033-5894. S2CID 129827927.


