Ceftriaxone
Ceftriaxone, sold under the brand name Rocephin, is a third-generation cephalosporin antibiotic used for the treatment of a number of bacterial infections.[3] These include middle ear infections, endocarditis, meningitis, pneumonia, bone and joint infections, intra-abdominal infections, skin infections, urinary tract infections, gonorrhea, and pelvic inflammatory disease.[3] It is also sometimes used before surgery and following a bite wound to try to prevent infection.[3] Ceftriaxone can be given by injection into a vein or into a muscle.[3]
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌsɛftraɪˈæksoʊn/ |
| Trade names | Rocephin, Epicephin, Wintriaxone, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a685032 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Intravenous, intramuscular |
| Drug class | Third-generation cephalosporin |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | n/a |
| Metabolism | Negligible |
| Elimination half-life | 5.8–8.7 hours[2] |
| Excretion | 33–67% kidney, 35–45% biliary |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.070.347 |
| Chemical and physical data | |
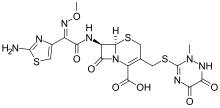
| Formula | C18H18N8O7S3 |
| Molar mass | 554.57 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Common side effects include pain at the site of injection and allergic reactions.[3] Other possible side effects include C. difficile-associated diarrhea, hemolytic anemia, gall bladder disease, and seizures.[3] It is not recommended in those who have had anaphylaxis to penicillin but may be used in those who have had milder reactions.[3] The intravenous form should not be given with intravenous calcium.[3] There is tentative evidence that ceftriaxone is relatively safe during pregnancy and breastfeeding.[1] It is a third-generation cephalosporin that works by preventing bacteria from making a cell wall.[3]
Ceftriaxone was patented in 1978 and approved for medical use in 1982.[4] It is on the World Health Organization's List of Essential Medicines.[5] It is available as a generic medication.[3]
Medical use
Ceftriaxone and other third-generation cephalosporin antibiotics are used to treat organisms that tend to be resistant to many other antibiotics.[6] Due to emergent resistance, ceftriaxone should not be used for the treatment of Enterobacter infections.[6] Before using ceftriaxone, it is important to determine the susceptibility of the bacteria.[7] If sepsis is being considered, empiric therapy may be initiated prior to susceptibility testing.[6]
Medical uses include:[7]
- lower respiratory tract infections
- acute bacterial otitis media
- skin and skin structure infections
- urinary tract infections
- uncomplicated gonorrhea
- pelvic inflammatory disease
- bacterial sepsis
- intra-abdominal infections
- meningitis
- surgical prophylaxis
- Lyme disease[3]
Ceftriaxone is also a choice drug for treatment of bacterial meningitis caused by pneumococci, meningococci, Haemophilus influenzae, and "susceptible enteric Gram-negative rods, but not Listeria monocytogenes."[8]
In combination with doxycycline or azithromycin, ceftriaxone used to be recommended by the United States Centers for Disease Control and Prevention (CDC) for the treatment of uncomplicated gonorrhea. Due to increased risk of developing azithromycin resistant strains and the high efficacy of higher doses of ceftriaxone the guidance has been updated to mono-antibiotic therapy with a higher dose of ceftriaxone.[9]
Spectrum of activity
Like other third-generation cephalosporins, ceftriaxone is active against Citrobacter spp., Serratia marcescens, and beta-lactamase-producing strains of Haemophilus and Neisseria.[6] However, unlike ceftazidime and cefoperazone, ceftriaxone does not have useful activity against Pseudomonas aeruginosa.[6] It is generally not active against Enterobacter species, and its use should be avoided in the treatment of Enterobacter infections, even if the isolate appears susceptible, because of the emergence of resistance.[6] Some organisms, such as Citrobacter, Providencia, and Serratia, have the ability to become resistant through the development of cephalosporinases (enzymes that hydrolyze cephalosporins and render them inactive).[6] Although not being used as first line therapy against Staphylococcus aures, ceftriaxone retains activity against isolates of methicillin-resistant S. aureus and is used in clinic for infections sustained by this bacterium. In this case the dose should be doubled (e.g. 2 g intravenously every 12 hours).[10]
Available forms
Ceftriaxone is available for administration via the intramuscular or the intravenous routes.[7] Diluents containing calcium should not be used to reconstitute ceftriaxone, and it must not be administered in intravenous lines containing other calcium-containing solutions, as a ceftriaxone-calcium precipitate could form.[7]
Pregnancy
Ceftriaxone is pregnancy category B.[7][1] It has not been observed to cause birth defects in animal studies, but a lack of well-controlled studies done in pregnant women exists.[7]
Breastfeeding
Low concentrations of ceftriaxone are excreted in breast milk that are "not expected to cause adverse effects in breastfed infants."[11] The manufacturer recommends that caution be exercised when administering ceftriaxone to women who breastfeed.[7]
Newborns
Hyperbilirubinemic neonates are contraindicated for the use of ceftriaxone.[7] It can compete with bilirubin and displace it from binding to albumin, increasing the risk of bilirubin encephalopathy.[7]
Elderly
According to the package insert, clinical studies did not show differences in efficacy and safety of ceftriaxone in geriatrics compared to younger patients but "greater sensitivity of some older individuals cannot be ruled out."[7]
Adverse effects
Although generally well tolerated, the most common adverse reactions associated with ceftriaxone are changes in white blood cell counts, local reactions at site of administration, rash, and diarrhea.[12]
Incidence of adverse effects greater than 1%:
- Eosinophilia (6%)
- Thrombocytosis (5.1%)
- Elevations in liver enzymes (3.1–3.3%)
- Diarrhea (2.7%)
- Leukopenia (2.1%)
- Elevation in BUN (1.2%)
- Local reactions: pain, tenderness, irritation (1%)
- Rash (1.7%)[12]
Some less frequently reported adverse events (incidence < 1%) include phlebitis, itchiness, fever, chills, nausea, vomiting, elevations of bilirubin, elevations in creatinine, headache and dizziness.[12]
Ceftriaxone may precipitate in bile, causing biliary sludge, biliary pseudolithiasis, and gallstones, especially in children. Hypoprothrombinaemia and bleeding are specific side effects. Haemolysis is reported.[13][14][15] It has also been reported to cause post kidney failure in children.[16] Like other antibiotics, ceftriaxone use can result in Clostridioides difficile-associated diarrhea ranging from mild diarrhea to fatal colitis.[12] In this regard it has been reported that shifting from ceftriaxone to cefotaxime would have a lower impact on C. difficile infection rates, since cefotaxime is almost entirely excreted by the kidneys [17] while ceftriaxone has a 45% biliary excretion [18]
Contraindications
Ceftriaxone should not be used in those with an allergy to ceftriaxone or any component of the formulation. Although there is negligible cross-reactivity between penicillins and third-generation cephalosporins,[8][19] caution should still be used when using ceftriaxone in penicillin-sensitive patients.[12] Caution should be used in people who have had previous severe penicillin allergies.[12] It should not be used in hyperbilirubinemic neonates, particularly those who are premature because ceftriaxone is reported to displace bilirubin from albumin binding sites, potentially causing bilirubin encephalopathy. Concomitant use with intravenous calcium-containing solutions/products in neonates (≤28 days) is contraindicated [20] even if administered through different infusion lines due to rare fatal cases of calcium-ceftriaxone precipitations in neonatal lungs and kidneys.[12][21]
Mechanism of action
Ceftriaxone is a third-generation antibiotic from the cephalosporin family of antibiotics.[6] It is within the β-lactam family of antibiotics. Ceftriaxone selectively and irreversibly inhibits bacterial cell wall synthesis by binding to transpeptidases, also called transamidases, which are penicillin-binding proteins (PBPs) that catalyze the cross-linking of the peptidoglycan polymers forming the bacterial cell wall.[22] The peptidoglycan cell wall is made up of pentapeptide units attached to a polysaccharide backbone with alternating units of N-acetylglucosamine and N-acetylmuramic acid.[23][24] PBPs act on a terminal D-alanyl-D-alanine moiety on a pentapeptide unit and catalyze the formation of a peptide bond between the penultimate D-alanine and a glycine unit on an adjacent peptidoglycan strand, releasing the terminal D-alanine unit in the process.[22][24] The structure of ceftriaxone mimics the D-alanyl-D-alanine moiety, and the PBP attacks the beta-lactam ring in ceftriaxone as if it were its normal D-alanyl-D-alanine substrate.[22] The peptidoglycan cross-linking activity of PBPs is a construction and repair mechanism that normally helps to maintain bacterial cell wall integrity, so the inhibition of PBPs leads to damage and destruction of the cell wall and eventually to cell lysis.[22]
Pharmacokinetics
Absorption: Ceftriaxone can be administered intravenously and intramuscularly, and the drug is completely absorbed.[7][25] It is not available orally.[26][27]
Distribution: Ceftriaxone penetrates tissues and body fluids well, including cerebrospinal fluid to treat central nervous system infections.[7][28] Ceftriaxone is reversibly bound to human plasma proteins and the binding of ceftriaxone decreases with increasing concentration from a value of 95% at plasma concentrations less than 25 mcg/mL to 85% at plasma concentration of 300 mcg/mL. Over a 0.15 to 3 g dose range in healthy adult subjects, the apparent volume of distribution ranged from 5.8 to 13.5 L.[7]
Metabolism: 33–67% of ceftriaxone is renally excreted as unchanged drug, but no dose adjustments are required in renal impairment with dosages up to 2 grams per day.[7] The rest[29] is excreted in the bile as unchanged drug[30] which is ultimately excreted in feces as inactive compounds from hepatic and gut flora metabolism.[7][31][32]
Elimination: The average elimination half-life in healthy adults is 5.8–8.7 (mean 6.5) hours,[2] with some reviews estimated half-life up to 10 hours.[33] In people with renal impairment, the average elimination half-life increases to 11.4–15.7 hours.[7]
Chemistry
Ceftriaxone is commercially available as a white to yellowish-orange crystalline powder for reconstitution.[7] Reconstituted ceftriaxone injection solutions are light yellow- to amber-colored depending on how long the solution had been reconstituted, the concentration of ceftriaxone in the solution, and the diluent used.[7] To reduce pain with intramuscular injections, ceftriaxone may be reconstituted with lidocaine.[34]
The syn-configuration of the methoxy oxime moiety confers resistance to beta-lactamase enzymes produced by many Gram-negative bacteria.[22] The stability of this configuration results in increased activity of ceftriaxone against otherwise resistant Gram-negative bacteria.[22] In place of the easily hydrolyzed acetyl group of cefotaxime, ceftriaxone has a metabolically stable thiotriazinedione moiety.[22]
Research
Ceftriaxone has also been investigated for efficacy in preventing relapse to cocaine addiction.[35]
Ceftriaxone seems to increase excitatory amino acid transporter-2 pump expression and activity in the central nervous system, so has a potential to reduce glutamatergic toxicity.[36][37]
Ceftriaxone has been shown to have neuroprotective properties in a number of neurological disorders, including spinal muscular atrophy[38] and amyotrophic lateral sclerosis (ALS).[39] Despite earlier negative results in the 1990s, a large clinical trial was undertaken in 2006 to test ceftriaxone in ALS patients, but was stopped early after it became clear that the results would not meet the predetermined criteria for efficacy.[40]
References
- "Ceftriaxone (Rocephin) Use During Pregnancy". Drugs.com. 12 December 2019. Archived from the original on 23 August 2016. Retrieved 24 December 2019.
- Beam TR (1985). "Ceftriaxone: a beta-lactamase-stable, broad-spectrum cephalosporin with an extended half-life". Pharmacotherapy. 5 (5): 237–253. doi:10.1002/j.1875-9114.1985.tb03423.x. PMID 3906584. S2CID 25559476.
- "Ceftriaxone Sodium Monograph for Professionals". Drugs.com. Archived from the original on 31 May 2016. Retrieved 27 August 2016.
- Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 495. ISBN 9783527607495.
- World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- Katzung B, Masters S, Trevor A (2012). Basic and Clinical Pharmacology. McGraw-Hill. pp. 797–801. ISBN 978-0-07-176402-5.
- "Ceftriaxone- ceftriaxone sodium injection, powder, for solution". DailyMed. 31 December 2019. Archived from the original on 17 November 2015. Retrieved 1 February 2020.
- Katzung B (2009). Basic and Clinical Pharmacology, Eleventh Edition. New York: McGraw-Hill. pp. 783–784. ISBN 978-0-07-160405-5.
- St Cyr S, Barbee L, Workowski KA, Bachmann LH, Pham C, Schlanger K, et al. (December 2020). "Update to CDC's Treatment Guidelines for Gonococcal Infection, 2020". MMWR. Morbidity and Mortality Weekly Report. 69 (50): 1911–1916. doi:10.15585/mmwr.mm6950a6. PMC 7745960. PMID 33332296.
- Di Bella S, Gatti M, Principe L (29 April 2023). "Ceftriaxone for methicillin-susceptible Staphylococcus aureus (MSSA) bacteremia: a matter of dosages?". European Journal of Clinical Microbiology & Infectious Diseases. 42 (7): 917–918. doi:10.1007/s10096-023-04612-x. ISSN 1435-4373. PMID 37119346. S2CID 258423746.
- "TOXNET". toxnet.nlm.nih.gov. Archived from the original on 8 September 2017. Retrieved 4 November 2015.
- "Rocephin Prescribing Information" (PDF). Roche. Archived (PDF) from the original on 4 March 2016. Retrieved 1 November 2015.
- Shiffman ML, Keith FB, Moore EW (December 1990). "Pathogenesis of ceftriaxone-associated biliary sludge. In vitro studies of calcium-ceftriaxone binding and solubility". Gastroenterology. 99 (6): 1772–1778. doi:10.1016/0016-5085(90)90486-K. PMID 2227290.
- Shrimali JD, Patel HV, Gumber MR, Kute VB, Shah PR, Vanikar AV, Trivedi HL (November 2013). "Ceftriaxone induced immune hemolytic anemia with disseminated intravascular coagulation". Indian Journal of Critical Care Medicine. 17 (6): 394–395. doi:10.4103/0972-5229.123465. PMC 3902580. PMID 24501497.
- Guleria VS, Sharma N, Amitabh S, Nair V (September–October 2013). "Ceftriaxone-induced hemolysis". Indian Journal of Pharmacology. 45 (5): 530–531. doi:10.4103/0253-7613.117758. PMC 3793531. PMID 24130395.
- Li N, Zhou X, Yuan J, Chen G, Jiang H, Zhang W (April 2014). "Ceftriaxone and acute renal failure in children". Pediatrics. 133 (4): e917–e922. doi:10.1542/peds.2013-2103. PMID 24664092. S2CID 2854637.
- Patel KB, Nicolau DP, Nightingale CH, Quintiliani R (May 1995). "Pharmacokinetics of cefotaxime in healthy volunteers and patients". Diagnostic Microbiology and Infectious Disease. 22 (1–2): 49–55. doi:10.1016/0732-8893(95)00072-I. PMID 7587050.
- Guggenbichler JP, Allerberger FJ, Dierich M (1986). "Influence of cephalosporines III generation with varying biliary excretion on fecal flora and emergence of resistant bacteria during and after cessation of therapy". Padiatrie und Padologie. 21 (4): 335–342. PMID 3562044. Archived from the original on 29 April 2023. Retrieved 29 April 2023.
- "The Use of Cephalosporins in Penicillin-allergic Patients". www.medscape.com. Archived from the original on 14 November 2015. Retrieved 10 November 2015.
- "FDA Updates warning on Ceftriaxone-Calcium injection". Archived from the original on 28 November 2009.
- Bradley JS, Wassel RT, Lee L, Nambiar S (April 2009). "Intravenous ceftriaxone and calcium in the neonate: assessing the risk for cardiopulmonary adverse events". Pediatrics. 123 (4): e609–e613. doi:10.1542/peds.2008-3080. PMID 19289450. S2CID 22718923.
- Lemke TL, Williams DA, eds. (2013). Foye's Principles of Medicinal Chemistry (Seventh ed.). Philadelphia, PA: Lippincott Williams & Wilkins. pp. 1093–1094, 1099–1100. ISBN 9781609133450.
- van Heijenoort J (March 2001). "Formation of the glycan chains in the synthesis of bacterial peptidoglycan". Glycobiology. 11 (3): 25R–36R. doi:10.1093/glycob/11.3.25r. PMID 11320055. S2CID 46066256.
- Scheffers DJ, Pinho MG (December 2005). "Bacterial cell wall synthesis: new insights from localization studies". Microbiology and Molecular Biology Reviews. 69 (4): 585–607. doi:10.1128/MMBR.69.4.585-607.2005. PMC 1306805. PMID 16339737.
- Patel IH, Kaplan SA (October 1984). "Pharmacokinetic profile of ceftriaxone in man". The American Journal of Medicine. 77 (4C): 17–25. PMID 6093513.
- Red Book: Pharmacy's Fundamental Reference (114th ed.). PDR Network, LLC. 2010. ISBN 9781563637513.
- "DailyMed – Search Results for ceftriaxone". dailymed.nlm.nih.gov. Archived from the original on 6 March 2016. Retrieved 4 November 2015.
- Nau R, Sörgel F, Eiffert H (October 2010). "Penetration of drugs through the blood-cerebrospinal fluid/blood-brain barrier for treatment of central nervous system infections". Clinical Microbiology Reviews. 23 (4): 858–883. doi:10.1128/CMR.00007-10. PMC 2952976. PMID 20930076.
- Arvidsson A, Alván G, Angelin B, Borgå O, Nord CE (September 1982). "Ceftriaxone: renal and biliary excretion and effect on the colon microflora". The Journal of Antimicrobial Chemotherapy. 10 (3): 207–215. doi:10.1093/jac/10.3.207. PMID 6292158.
- Blumer J (September 1991). "Pharmacokinetics of ceftriaxone". Hospital Practice. 26 (Suppl 5): 7–13, discussion 52–4. doi:10.1080/21548331.1991.11707737. PMID 1918224.
- Balant L, Dayer P, Auckenthaler R (1 April 1985). "Clinical pharmacokinetics of the third generation cephalosporins". Clinical Pharmacokinetics. 10 (2): 101–143. doi:10.2165/00003088-198510020-00001. PMID 3888488. S2CID 23478077.
- Nursing Pharmacology Made Incredibly Easy!. Lippincott Williams & Wilkins. 7 March 2012. p. 496. ISBN 9781451146240. Archived from the original on 3 June 2016.
- Klein NC, Cunha BA (July 1995). "Third-generation cephalosporins". The Medical Clinics of North America. 79 (4): 705–719. doi:10.1016/s0025-7125(16)30034-7. PMID 7791418.
- Schichor A, Bernstein B, Weinerman H, Fitzgerald J, Yordan E, Schechter N (January 1994). "Lidocaine as a diluent for ceftriaxone in the treatment of gonorrhea. Does it reduce the pain of the injection?". Archives of Pediatrics & Adolescent Medicine. 148 (1): 72–75. doi:10.1001/archpedi.1994.02170010074017. PMID 8143016.
- Knackstedt LA, Melendez RI, Kalivas PW (January 2010). "Ceftriaxone restores glutamate homeostasis and prevents relapse to cocaine seeking". Biological Psychiatry. 67 (1): 81–84. doi:10.1016/j.biopsych.2009.07.018. PMC 2795043. PMID 19717140.
- Verma R, Mishra V, Sasmal D, Raghubir R (July 2010). "Pharmacological evaluation of glutamate transporter 1 (GLT-1) mediated neuroprotection following cerebral ischemia/reperfusion injury". European Journal of Pharmacology. 638 (1–3): 65–71. doi:10.1016/j.ejphar.2010.04.021. PMID 20423712.
- Lee SG, Su ZZ, Emdad L, Gupta P, Sarkar D, Borjabad A, et al. (May 2008). "Mechanism of ceftriaxone induction of excitatory amino acid transporter-2 expression and glutamate uptake in primary human astrocytes". The Journal of Biological Chemistry. 283 (19): 13116–13123. doi:10.1074/jbc.M707697200. PMC 2442320. PMID 18326497.
- Hedlund E (September 2011). "The protective effects of β-lactam antibiotics in motor neuron disorders". Experimental Neurology. 231 (1): 14–18. doi:10.1016/j.expneurol.2011.06.002. PMID 21693120. S2CID 26353910.
- Rothstein JD, Patel S, Regan MR, Haenggeli C, Huang YH, Bergles DE, et al. (January 2005). "Beta-lactam antibiotics offer neuroprotection by increasing glutamate transporter expression". Nature. 433 (7021): 73–77. Bibcode:2005Natur.433...73R. doi:10.1038/nature03180. PMID 15635412. S2CID 4301666.
- "Statement on the Clinical Trial of Ceftriaxone". The Northeast ALS Consortium (NEALS). 8 August 2012. Archived from the original on 28 May 2013. Retrieved 10 May 2013.
External links
- "Ceftriaxone". Drug Information Portal. U.S. National Library of Medicine.